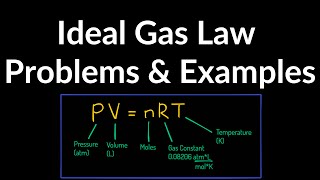
21. Kinetic Theory of Ideal Gases
The Ideal Gas Law
Multiple Choice
Multiple ChoiceHydrogen gas behaves very much like an ideal gas. If you have a sample of Hydrogen gas with a volume of 1000 cm3 at 30°C with a pressure of 1 × 105 Pa, calculate how many hydrogen atoms (particles) there are in the sample.
A
2.42 × 1022
B
2.39 × 1022
C
2.39 × 1028
1275
views
3
rank
Related Videos
Related Practice
Showing 1 of 10 videos