21. Kinetic Theory of Ideal Gases
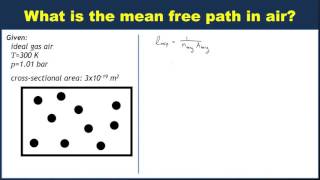
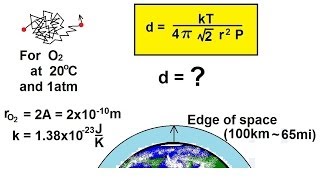
Mean Free Path of Gases
Problem 20b
Textbook Question
Textbook QuestionPhotons of light scatter off molecules, and the distance you can see through a gas is proportional to the mean free path of photons through the gas. Photons are not gas molecules, so the mean free path of a photon is not given by Equation 20.3, but its dependence on the number density of the gas and on the molecular radius is the same. Suppose you are in a smoggy city and can barely see buildings 500 m away. a. How far would you be able to see if all the molecules around you suddenly doubled in volume?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
300
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos