20. Heat and Temperature
Advanced Calorimetry: Equilibrium Temperature with Phase Changes
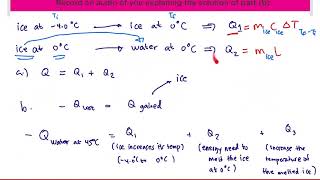
Problem 19.28
Textbook Question
Textbook Question(II) Determine the latent heat of fusion of mercury using the following calorimeter data: 1.00 kg of solid Hg at its melting point of −39.0°C is placed in a 0.620-kg aluminum calorimeter with 0.400 kg of water at 12.80°C; the resulting equilibrium temperature is 5.06°C.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
8mPlay a video:
65
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 8 videos