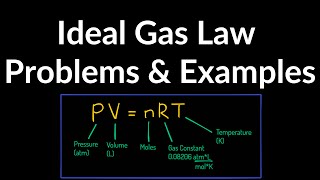
21. Kinetic Theory of Ideal Gases
The Ideal Gas Law
Problem 19b
Textbook Question
Textbook QuestionA 6.0-cm-diameter cylinder of nitrogen gas has a 4.0-cm-thick movable copper piston. The cylinder is oriented vertically, as shown in FIGURE P19.49, and the air above the piston is evacuated. When the gas temperature is 20°C, the piston floats 20 cm above the bottom of the cylinder. c. What is the new equilibrium temperature of the gas?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
0m:0sPlay a video:
183
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos