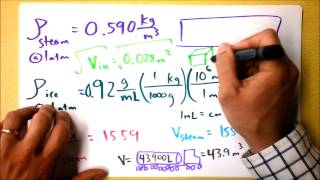20. Heat and Temperature
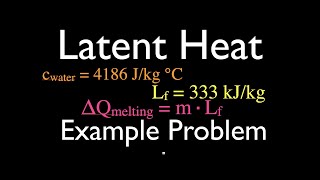
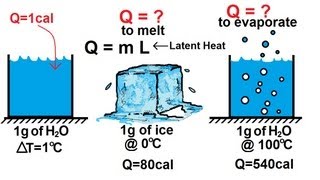
Latent Heat & Phase Changes
Problem 19b
Textbook Question
Textbook QuestionWhen air is inhaled, it quickly becomes saturated with water vapor as it passes through the moist airways. Consequently, an adult human exhales about 25 mg of evaporated water with each breath. Evaporation—a phase change—requires heat, and the heat energy is removed from your body. Evaporation is much like boiling, only water's heat of vaporization at 35°C is a somewhat larger 24×10^5 J/kg because at lower temperatures more energy is required to break the molecular bonds. At 12 breaths/min, on a dry day when the inhaled air has almost no water content, what is the body's rate of energy loss (in J/s) due to exhaled water? (For comparison, the energy loss from radiation, usually the largest loss on a cool day, is about 100 J/s.)
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
277
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos