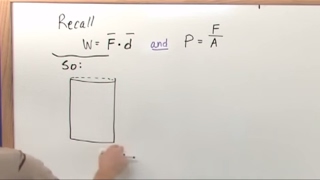22. The First Law of Thermodynamics
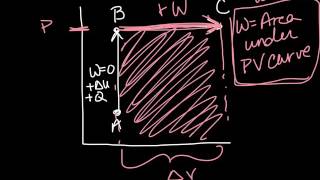
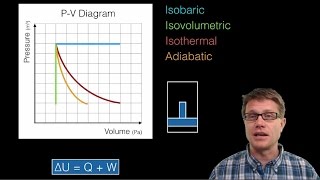
PV Diagrams & Work
Problem 19.38b
Textbook Question
Textbook Question(II) A 1.0-L volume of air initially at 3.5 atm of (gauge)pressure is allowed to expand isothermally until the (gauge) pressure is 1.0 atm. It is then compressed at constant pressure to its initial volume, and lastly is brought back to its original pressure by heating at constant volume.
(b) How much work does the 1.0 L of air do in this process?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
12mPlay a video:
43
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos