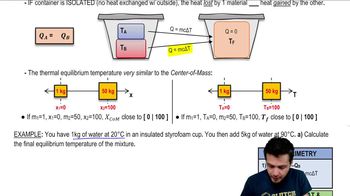
20. Heat and Temperature
Intro to Calorimetry
Problem 17c
Textbook Question
Textbook QuestionA copper pot with a mass of 0.500 kg contains 0.170 kg of water, and both are at 20.0°C. A 0.250-kg block of iron at 85.0°C is dropped into the pot. Find the final temperature of the system, assuming no heat loss to the surroundings.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
8mPlay a video:
1005
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 11 videos