21. Kinetic Theory of Ideal Gases
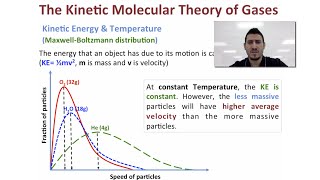
Kinetic-Molecular Theory of Gases
Problem 18.41
Textbook Question
Textbook Question(II) In the van der Waals equation of state, the constant b represents the amount of “unavailable volume” (per mole) occupied by the molecules themselves. Thus V is replaced by (V - nb) , where n is the number of moles. For oxygen, b is about 3.2 x 10⁻⁵ m³ /mol . Estimate the diameter of an oxygen molecule.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
6mPlay a video:
68
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 8 videos