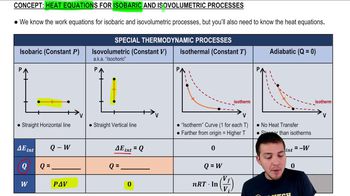
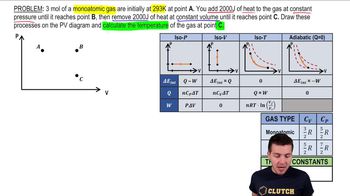
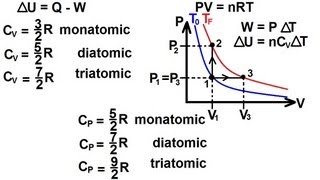
22. The First Law of Thermodynamics
Heat Equations for Special Processes & Molar Specific Heats
Problem 19.92
Textbook Question
Textbook QuestionSuppose 3.0 mol of neon (a monatomic gas, assume ideal) at STP are compressed slowly and isothermally to 0.22 the original volume. The gas is then allowed to expand quickly and adiabatically back to its original volume. Find the highest and lowest temperatures and pressures attained by the gas, and show on a PV diagram where these values occur.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
13mPlay a video:
78
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos