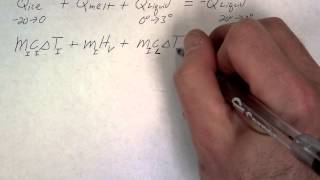20. Heat and Temperature
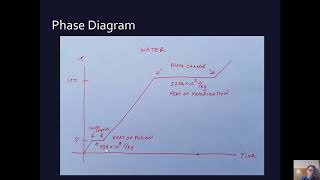
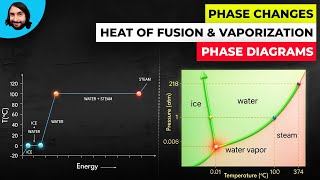
Calorimetry with Temperature and Phase Changes
Problem 19.24
Textbook Question
Textbook Question(II) A 28-g ice cube at its melting point is dropped into an insulated container of liquid nitrogen. How much nitrogen evaporates if it is at its boiling point of 77 K and has a latent heat of vaporization of 200 kJ/kg? Assume for simplicity that the specific heat of ice is a constant and is equal to its value near its melting point.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
85
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos