21. Kinetic Theory of Ideal Gases
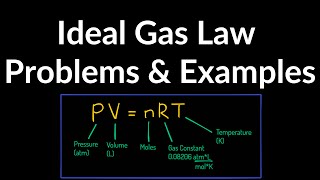
The Ideal Gas Law
Problem 18.74
Textbook Question
Textbook QuestionUsing the ideal gas law, find an expression for the mean free path ℓ_M that involves pressure and temperature instead of (N/V). Use this expression to find the mean free path for nitrogen molecules at a pressure of 7.5 atm and 300 K.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
71
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos