21. Kinetic Theory of Ideal Gases
The Ideal Gas Law
Problem 18c
Textbook Question
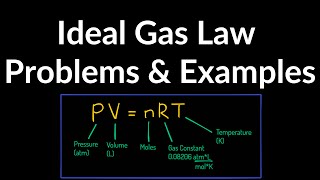
Textbook QuestionHelium gas with a volume of 3.20 L, under a pressure of 0.180 atm and at 41.0°C, is warmed until both pressure and volume are doubled. (b) How many grams of helium are there? The molar mass of helium is 4.00 g/mol.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
5mPlay a video:
458
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos