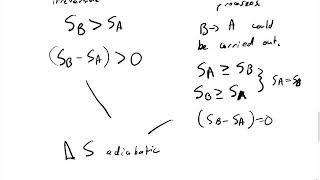23. The Second Law of Thermodynamics
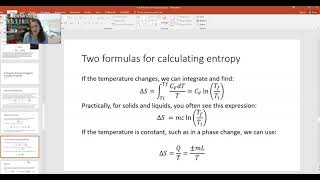
Entropy Equations for Special Processes
Multiple Choice
Multiple Choice3 moles of an ideal gas are in the left side of an hourglass-shaped container, separated by a thin barrier. The right side is completely empty, but the volume of the left and right sides are equal. The barrier is suddenly removed, and the gas freely expands into the vacuum. What is the change in entropy?
A
17.3 J/K
B
0 J/K
C
–17.3 J/K
D
49.9 J/K
299
views
1
comments
Related Videos
Related Practice
Showing 1 of 10 videos