22. The First Law of Thermodynamics
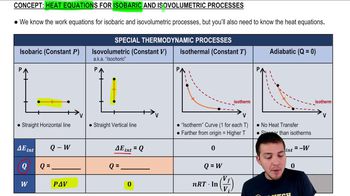
Heat Equations for Special Processes & Molar Specific Heats
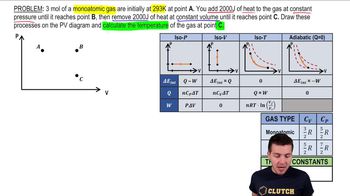
Problem 19.53
Textbook Question
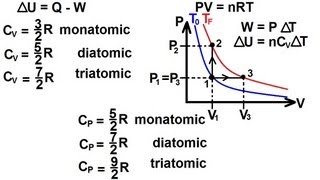
Textbook Question(II) Show that the work done by n moles of an ideal gas when it expands adiabatically is W = nC_v( T₁ - T₂) , where T₁ and T₂ are the initial and final temperatures, and C_v is the molar specific heat at constant volume.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
66
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos