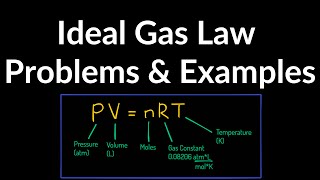
21. Kinetic Theory of Ideal Gases
The Ideal Gas Law
Textbook Question
Textbook Question(III) Estimate how many air molecules rebound from a wall in a typical room per second, assuming an ideal gas of N molecules contained in a cubic room with sides of length ℓ at temperature T and pressure P.
(a) Show that the frequency f with which gas molecules strike a wall is ƒ = (υ¯ₓ /2)(P/kT) ℓ² where υ¯ₓ is the average x component of the molecule’s velocity.
(b) Show that the equation can then be written as ƒ≈ Pℓ² /√4mkT where m is the mass of a gas molecule.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
9mPlay a video:
60
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos