23. The Second Law of Thermodynamics
Entropy Equations for Special Processes
Problem 20.92
Textbook Question
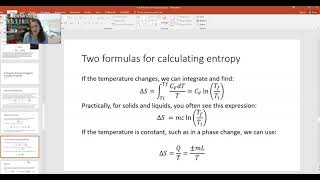
Textbook QuestionAt low temperature the specific heat of diamond varies with absolute temperature T according to the Debye equation C_V = 1.88 x 10³ (T/T_D)³ J•mol⁻¹ • K⁻¹ where the Debye temperature for diamond is T_D = 2230 K . Determine the entropy change of 1.00 mol of diamond when it is heated at constant volume from 4 K to 40 K. [Hint: dS = nC _VdT/T , where n is the number of moles.]
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
8mPlay a video:
38
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos