22. The First Law of Thermodynamics
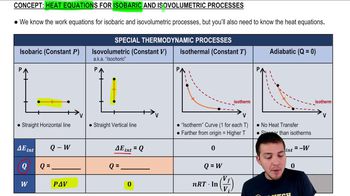
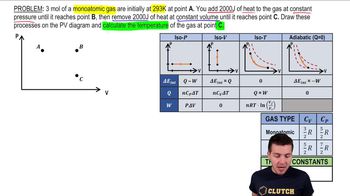
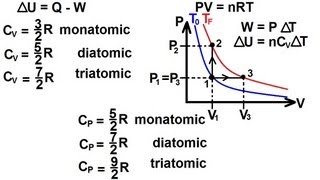
Heat Equations for Special Processes & Molar Specific Heats
Problem 19.95
Textbook Question
Textbook QuestionAt very low temperatures, the molar specific heat of many substances varies as the cube of the absolute temperature:
C = k (T³ / T³₀)
which is sometimes called Debye’s law. For rock salt, T₀ = 281 K and k = 1940 J/mol · K. Determine the heat needed to raise 2.75 mol of salt from 22.0 K to 46.0 K.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
71
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos