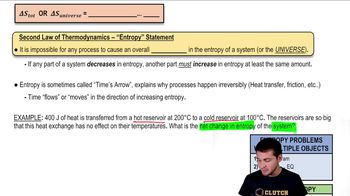
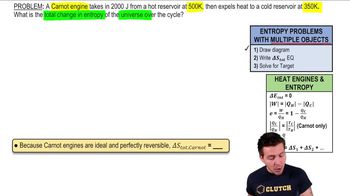
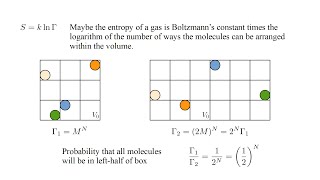
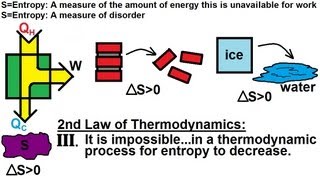
23. The Second Law of Thermodynamics
Entropy and the Second Law of Thermodynamics
Multiple Choice
Multiple ChoiceYou have a block of ice at 0°C. Heat is added to the ice, causing an increase in entropy of 120J/K. How much ice melts into water in this process?
A
0.098 kg
B
0 kg
C
1.32×10-6 kg
D
1.47×105 kg
444
views
3
rank
Related Videos
Related Practice
Showing 1 of 11 videos