22. The First Law of Thermodynamics
Work Done Through Multiple Processes
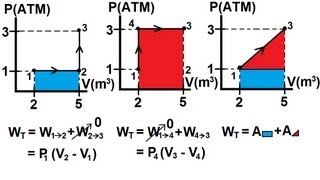
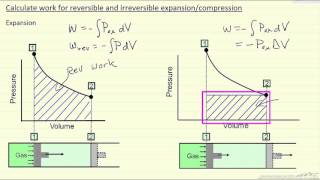
Problem 19c
Textbook Question
Textbook QuestionA gas undergoes two processes. In the first, the volume remains constant at 0.200 m^3 and the pressure increases from 2.00 * 10^5 Pa to 5.00 * 10^5 Pa. The second process is a compression to a volume of 0.120 m^3 at a constant pressure of 5.00 * 10^5 Pa. (b) Find the total work done by the gas during both processes.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
6mPlay a video:
951
views
2
rank
Was this helpful?
Related Videos
Related Practice
Showing 1 of 8 videos