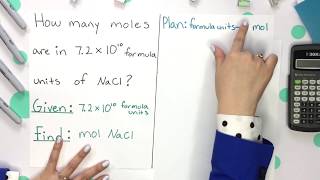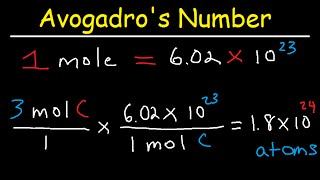20. Heat and Temperature
Moles and Avogadro's Number
Problem 17.42
Textbook Question
Textbook Question(II) A tank contains 30.0 kg of O₂ gas at a gauge pressure of 8.20 atm. If the oxygen is replaced by helium at the same temperature, how many kilograms of the latter will be needed to produce a gauge pressure of 8.00 atm?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
6mPlay a video:
84
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos