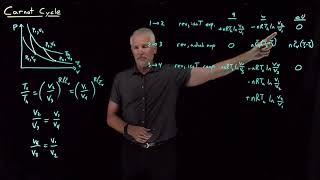23. The Second Law of Thermodynamics
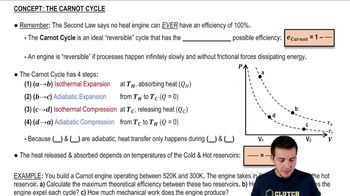
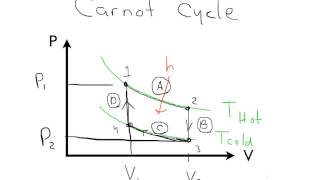
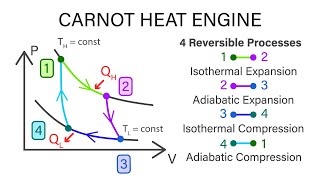
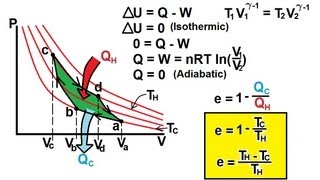
The Carnot Cycle
Problem 20.18
Textbook Question
Textbook Question(II) The working substance of a certain Carnot engine is 1.0 mol of an ideal monatomic gas. During the isothermal expansion portion of this engine’s cycle, the volume of the gas doubles, while during the adiabatic expansion the volume increases by a factor of 6.2. The work output of the engine is 920 J in each cycle. Compute the temperatures of the two reservoirs between which this engine operates.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
15mPlay a video:
67
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos