23. The Second Law of Thermodynamics
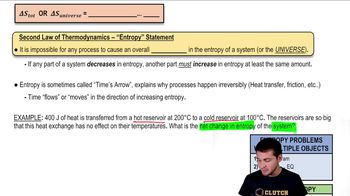
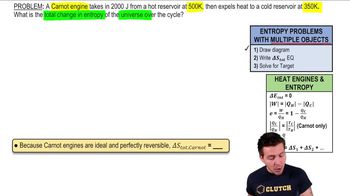
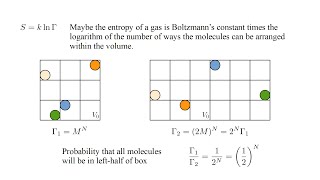
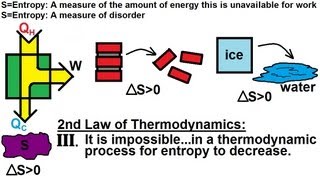
Entropy and the Second Law of Thermodynamics
Problem 20f
Textbook Question
Textbook QuestionA 75 g ice cube at 0℃ is placed on a very large table at 20℃. You can assume that the temperature of the table does not change. As the ice cube melts and then comes to thermal equilibrium, what are the entropy changes of (a) the water, (b) the table, and (c) the universe?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
16mPlay a video:
232
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 11 videos