22. The First Law of Thermodynamics
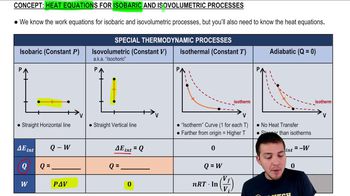
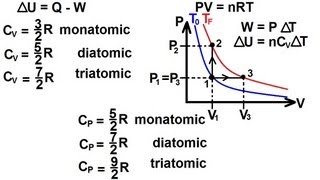
Heat Equations for Special Processes & Molar Specific Heats
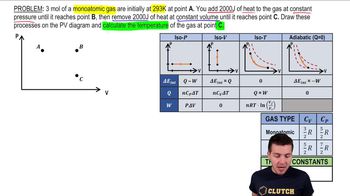
Problem 19x
Textbook Question
Textbook QuestionAn experimenter adds 970 J of heat to 1.75 mol of an ideal gas to heat it from 10.0°C to 25.0°C at constant pressure. The gas does +223 J of work during the expansion. (b) Calculate γ for the gas.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
6mPlay a video:
366
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos