21. Kinetic Theory of Ideal Gases
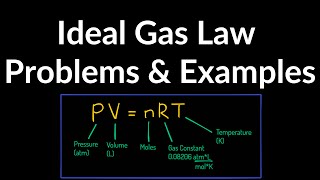
The Ideal Gas Law
Problem 20.76a
Textbook Question
Textbook Question1.00 mole of an ideal monatomic gas at STP first undergoes an isothermal expansion so that the volume at b is 2.5 times the volume at a (Fig. 20–25). Next, heat is extracted at a constant volume so that the pressure drops. The gas is then compressed adiabatically back to the original state.
(a) Calculate the pressures at b and c.
<IMAGE>
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
6mPlay a video:
75
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos