22. The First Law of Thermodynamics
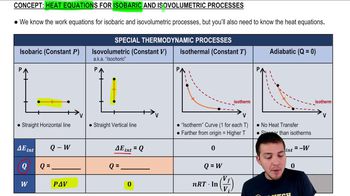
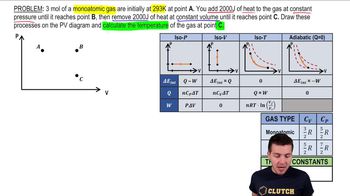
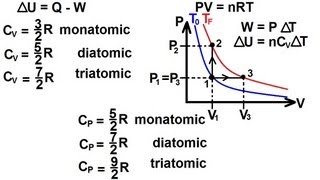
Heat Equations for Special Processes & Molar Specific Heats
Problem 19.56
Textbook Question
Textbook Question(II) A 2.00-mole sample of N₂ gas at 0°C is heated to 150°C at constant pressure (1.00 atm). Determine (a) the change in internal energy, (b) the work the gas does, and (c) the heat added to it.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
10mPlay a video:
42
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 9 videos