21. Kinetic Theory of Ideal Gases
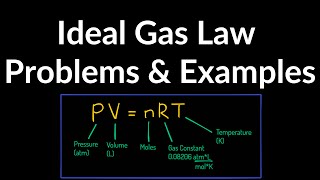
The Ideal Gas Law
Problem 18~
Textbook Question
Textbook Question10 g of dry ice (solid CO₂) is placed in a 10,000 cm^3 container, then all the air is quickly pumped out and the container sealed. The container is warmed to 0°C, a temperature at which CO₂ is a gas. a. What is the gas pressure? Give your answer in atm. The gas then undergoes an isothermal compression until the pressure is 3.0 atm, immediately followed by an isobaric compression until the volume is 1000 cm^3.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
6mPlay a video:
323
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos