21. Kinetic Theory of Ideal Gases
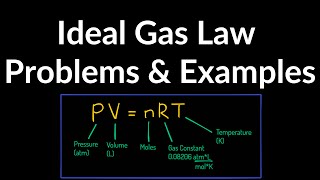
The Ideal Gas Law
Problem 18
Textbook Question
Textbook QuestionA diving bell is a 3.0-m-tall cylinder closed at the upper end but open at the lower end. The temperature of the air in the bell is 20°C. The bell is lowered into the ocean until its lower end is 100 m deep. The temperature at that depth is 10°C. a. How high does the water rise in the bell after enough time has passed for the air inside to reach thermal equilibrium?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
13mPlay a video:
286
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos