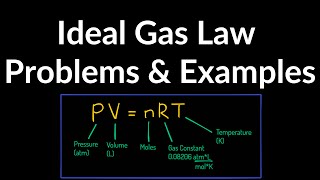
21. Kinetic Theory of Ideal Gases
The Ideal Gas Law
Problem 18r
Textbook Question
Textbook QuestionHelium gas with a volume of 3.20 L, under a pressure of 0.180 atm and at 41.0°C, is warmed until both pressure and volume are doubled. (a) What is the final temperature?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
4mPlay a video:
507
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos