20. Heat and Temperature
Advanced Calorimetry: Equilibrium Temperature with Phase Changes
Problem 17.41
Textbook Question
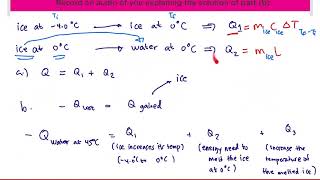
Textbook QuestionA 6.00-kg piece of solid copper metal at an initial temperature T is placed with 2.00 kg of ice that is initially at -20.0°C. The ice is in an insulated container of negligible mass and no heat is exchanged with the surroundings. After thermal equilibrium is reached, there is 1.20 kg of ice and 0.80 kg of liquid water. What was the initial temperature of the piece of copper?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
9mPlay a video:
36
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 8 videos