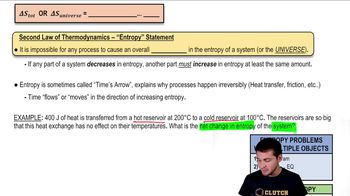
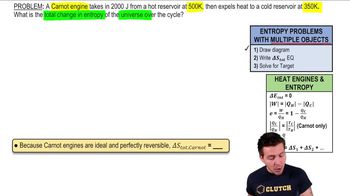
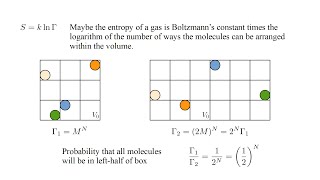
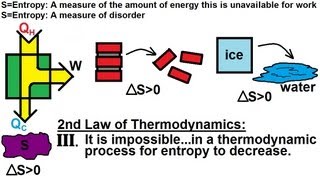
23. The Second Law of Thermodynamics
Entropy and the Second Law of Thermodynamics
Problem 20.51b
Textbook Question
Textbook Question(II) Two samples of an ideal gas are initially at the same temperature and pressure. They are each compressed reversibly from a volume V to volume V/2, one isothermally, the other adiabatically.
(b) Determine the change in entropy of the gas for each process by integration.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
6mPlay a video:
68
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 11 videos