21. Kinetic Theory of Ideal Gases
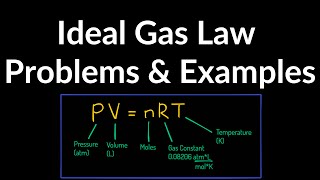
The Ideal Gas Law
Problem 18.77a
Textbook Question
Textbook Question(a) From the van der Waals equation of state, show that the critical temperature and pressure are given by
T꜀ᵣ = 8a / 27bR , P꜀ᵣ = a / 27b² .
[Hint: Use the fact that the P versus V curve has an inflection point at the critical point so that the first and second derivatives are zero.]
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
12mPlay a video:
71
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 10 videos