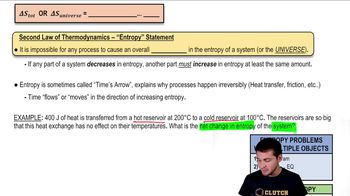
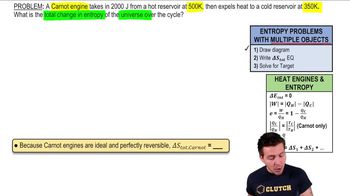
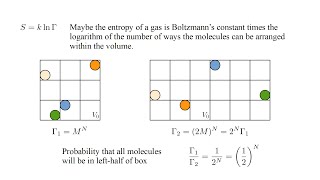
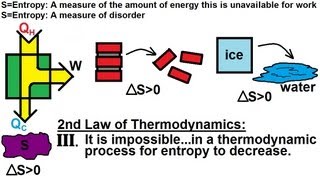
23. The Second Law of Thermodynamics
Entropy and the Second Law of Thermodynamics
Problem 20.38d
Textbook Question
Textbook Question(II) If 0.45 kg of water at 100°C is changed by a reversible process to steam at 100°C, determine the change in entropy of the water, the surroundings, and the universe as a whole.
(d) How would your answers differ if the process were irreversible?
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
8mPlay a video:
72
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 11 videos