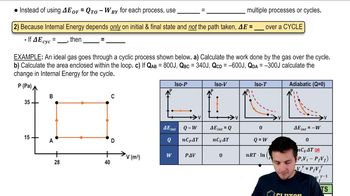
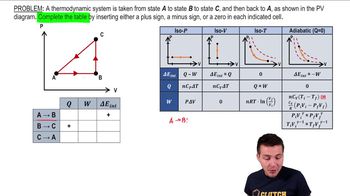
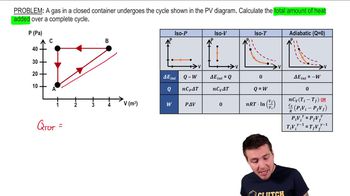
22. The First Law of Thermodynamics
Cyclic Thermodynamic Processes
Problem 19.42
Textbook Question
Textbook Question(II) An ideal gas expands at a constant total pressure of 2.5 atm from 410 mL to 690 mL. Heat then flows out of the gas at constant volume, and the pressure and temperature are allowed to drop until the temperature reaches its original value. Calculate
(a) the total work done by the gas in the process, and
(b) the total heat flow into the gas.
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
7mPlay a video:
69
views
Was this helpful?
Related Videos
Related Practice