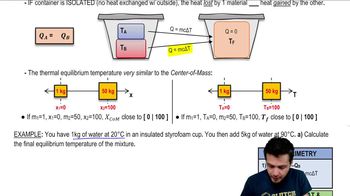
20. Heat and Temperature
Intro to Calorimetry
Problem 19.18
Textbook Question
Textbook Question(II) A 215-g sample of a substance is heated to 330°C and then plunged into a 105-g aluminum calorimeter cup containing 185 g of water and a 17-g glass thermometer at 10.5°C. The final temperature is 35.0°C. What is the specific heat of the substance? (Assume no water boils away.)
 Verified Solution
Verified SolutionThis video solution was recommended by our tutors as helpful for the problem above
Video duration:
9mPlay a video:
67
views
Was this helpful?
Related Videos
Related Practice
Showing 1 of 11 videos