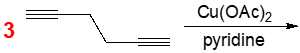In this video, we're going to take a look at the Eglinton reaction. Now, we're going to say that the Eglinton coupling reaction involves the coupling between two identical terminal alkynes with a copper catalyst and base. We want to make sure that the two alkynes we're using are identical so as to avoid a mixture of possible products. We're going to say here that the reaction uses a copper catalyst in the formation of a bialkynal product. And like coupling reactions, it has the same two driving forces. One driving force is the formation of conjugated products. Remember, the more conjugation our compound possesses, the more stable it is. And, we're going to say that another driving force is that unlike other coupling reactions, it doesn't use a catalytic cycle but instead radicals. The general setup for an Eglinton coupling reaction is that we have alkyne 1, which is identical to alkyne 2. We utilize copper 1 and copper 2 catalysts. And we have pyridine as our base. Remember, pyridine is like benzene except there's a nitrogen within it. And basically, what happens here is that we have the loss of hydrogen from both of these so that the alkyne portions that are left at the end combine together to give us our bialkynal product. We're going to look at the mechanism later on to see how it really occurs. But for simplicity's sake, you could just think of it as the two hydrogens being lost, and what is left combines together. Since our two alkynes are identical, that means that their R groups would be the same. So here we're going to say the R1 and R2 groups of the terminal alkynes can be represented by vinyl or aryl groups. They can also be represented by an alkyl group or even another alkyl group. So that's just the basic setup of the Eglinton reaction. Now that we've talked about the basic steps involved, click on to the next video and see how I approach the example question where we are asked to find the final product.
Eglinton Reaction - Online Tutor, Practice Problems & Exam Prep
Eglinton Reaction
Video transcript
Eglinton Reaction Example 1
Video transcript
Although we haven't gone over the coupling mechanism for the Eglinton coupling reaction, we can take a simple approach to find our answer. So if we take a look at this example here, it says determine the product from the following Eglinton coupling reaction. Now, here we have 2 identical terminal alkynes. If this were an actual question, you wouldn't have the number 2 here given to you. You'd have to realize that, oh, I'm dealing with the Eglinton coupling reaction. Therefore, I'm dealing with 2 identical terminal alkynes. So it's kind of implied that there isn't just one of these alkynes, there's actually 2 of them. Now all we have to do is, we're going to draw 1 terminal alkyne, then realize that there's going to be an identical one also involved in the reaction. And in terms of simplicity, we know that through a process that we haven't seen yet, that the hydrogens that are on the terminal alkyne carbons will be lost somehow and that the alkyne carbons then would connect with one another. This would give me my final product as, okay. We'd have this terminal alkyne connecting with this terminal alkyne. So this would be our final product. Now, this reaction also involves copper 2 acetate being used here and we have pyridine base also involved in this reaction. But again, even though we haven't done the mechanism yet for this coupling reaction, we know that from what we've seen up above, the hydrogens on both ends of the terminal alkynes would have been lost and so those 2 carbons left behind would connect together to give us our bialkynyl product that we have here. Now that we've seen this simple example question, click on to the next video and see how exactly does the coupling mechanism work for the Eglinton coupling reaction.
Eglinton Reaction
Video transcript
So, the Eglinton coupling reaction can be seen as being comprised of 4 basic steps when it comes to its coupling mechanism. Step 1 involves deprotonation. Step 2 substitution. Step 3 is radicalization and finally step 4 is dimerization. Now, if we start out with deprotonation, we're going to say the slight acidity of the terminal alkyne hydrogen allows it to be deprotonated by the pyridine base. So here we have this slightly acidic hydrogen. Remember that the nitrogen within our pyridine base has a lone pair. It utilizes that lone pair to remove this hydrogen here. When it's removed, carbon holds on to the electrons within the bond. So as a result, we create an alkynide ion. Plus, as a byproduct, we make Pyridium ion as our second product. Now byproduct, we're not really going to be concerned with. Now we head into step 2, substitution. So the alkynide ion that we made in step 1, it's formed during deprotonation undergoes a substitution with copper acetate. So here, this carbon uses its lone pair to attach to carbon which causes the breaking of this bond towards the acetate ion. So, we wind up making is we have our alkyne here that is now connected to this copper. And we have as another byproduct our acetate ion. From here we go into the 3rd step which is radicalization. So the newly formed carbon-copper bond undergoes homolytic cleavage in order to form an alkynide radical. So remember, in homolytic cleavage we have equal splitting of our single bond. So this bond will split. The terminal alkyne carbon will hold on to an electron. This carbon this copper here uses this electron to actually attach itself to this acetate. Now remember, with homolytic cleavage we don't have full arrows. Instead we have half or hook arrows being used because we only have the movement of individual electrons. At the same time that this acetate uses this electron to connect to this copper, this copper in the bond takes its electron and holds on to it. So as a result, what we'll have at this point is our alkynide radical, plus 2, copper's connected to acetates. Now, finally we have dimerization. So the final step involves the dimerization of the 2 alkynide radicals that have been formed. Notice I say 2 alkynide radicals. That's because during this entire process, remember, for this coupling reaction, we're using 2 identical terminal alkynes. So all that's happening here to alkyne number 1, the same exact thing has been happening to alkyne number 2, which is why by the time we get to our 4th step, we have 2 alkynide radicals. Dimerization can be seen as a form of termination where 2 radicals join together to form a new stable compound. So here, half arrow because we're moving 1 electron, half arrow because we're moving 1 electron, and now these 2 are joined together. So this would be our final product. Coupling reactions, that you might be used to. It doesn't use a catalytic cycle in order to form the product, instead we have the utilization of radicals. As long as you can remember the basic setup for this type of coupling reaction, you can get to the product very easily. But if you need to work out the mechanism, just remember that it's comprised of 4 steps within its mechanism. We have deprotonation, substitution, radicalization, and then finally dimerization to get our bialkenal product.
Determine compounds A and B from the following reaction sequence.

Problem Transcript
Predict the product formed from the following intramolecular Eglinton reaction.