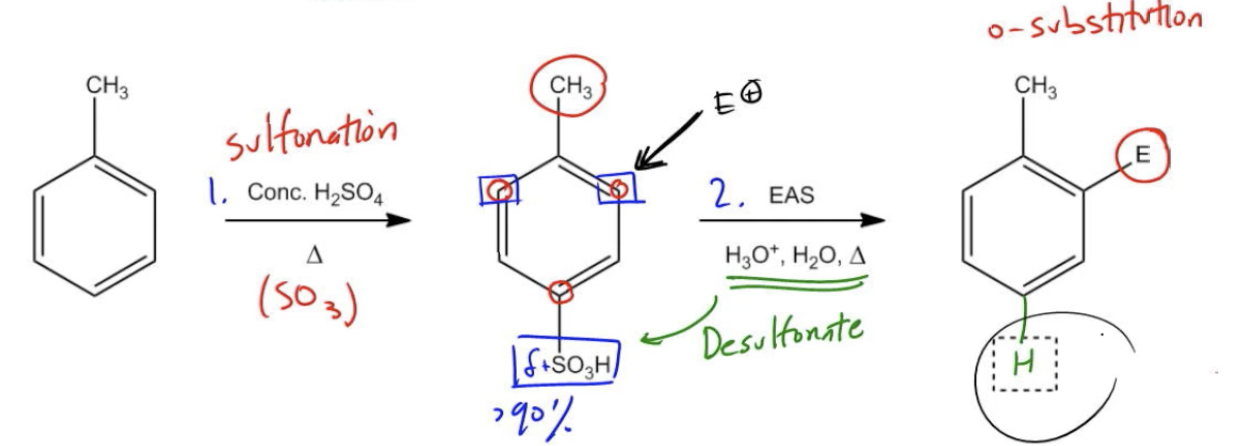
In the first step, I'm going to sulfonate. That sulfonation reaction is going to produce a sulfonic acid group. It's going to be para because like I told you guys, you get very high yields of the para product because para is favored over ortho. Now we're going to nitrate. Concentrated HNO3 is a nitration reaction. So now what we're going to do is we're going to get nitration. And where is it going to add? Well guys, it has to add here. Let's try to use a different color. It has to add here because the OH is still an ortho-para director. It's still going to direct ortho-para but it can't add para anymore because there's something there already. Then you have to add ortho. Then as you guys know, dilute acid heat is going to desulfonate and I'm going to end up with my final product that looks like this, OH and NO2. So I get my ortho substitution. I know there might be a few of you out there wondering, Johnny, if you did the nitration straight on phenol, wouldn't the major product be ortho anyway? I actually talked about how this is one of the exceptions; how you can get hydrogen bonding, so it actually favors ortho. You're exactly right and I'm glad you've been paying such close attention. But the problem is it still wouldn't be that great. It'll be like 60% if you just did it by itself. If all you did was you did concentrated HNO3, right? What you would get is a phenyl with a nitro group in about a 60% yield. But if you do this 3-step pathway, it's not much harder. You're going to get a 90% plus yield. See what I mean? Even in a situation where it would have been favored to go ortho, it's still better to use a blocking group so you can get a higher yield blocking that para position. Does that make more sense now? Awesome guys. Great example. Let's move on to the next topic.
19. Reactions of Aromatics: EAS and Beyond
Blocking Groups - Sulfonic Acid
19. Reactions of Aromatics: EAS and Beyond
Blocking Groups - Sulfonic Acid - Online Tutor, Practice Problems & Exam Prep
Let's discuss the one reversible EAS reaction and how we could use it to benefit us, specifically by acting as a blocking group.
1
concept
Forcing Ortho substitution
Video duration:
4mPlay a video:
2
example
Multistep Synthesis
Video duration:
2mPlay a video:
Video transcript

3
Problem
ProblemBeginning from Benzene, synthesize the following compound.

Video duration:
5mPlay a video:
Was this helpful?
Problem Transcript
Do you want more practice?
More setsYour Organic Chemistry tutors
Additional resources for Blocking Groups - Sulfonic Acid
PRACTICE PROBLEMS AND ACTIVITIES (1)

