This is a very typical multi-step synthesis problem that you would face in this chapter. There's a lot going on. The first thing that we notice is that first of all, none of the substituents on the first molecule are on the second. We either have to figure out how to remove them or how to transform them. In the case of the nitro, do we know any ways to transform a nitro group into an amino group? Hell yeah. We do. We'll come back to that in a second. Now we are also adding a chlorine. Do we now have the tools to add chlorine to a benzene ring? Yep, that's Electrophilic Aromatic Substitution (EAS). You might be wondering why this takes 4 steps. If you're getting aniline, if you're working with aniline, you're going to have to protect it. Let's start off at the beginning. What do you think the first reagent should be? For example, can my first reagent be an EAS chlorination? Should I do that first? No, guys, you can't because if you chlorinate right now, you have a meta director. You're going to wind up getting a chlorine here. Is that what we want? No. We need to transform it to an ortho/para director first, meaning that I have to reduce it first. Now you could use any of the reducing agents that I've mentioned in the past. All of them will be correct. I'm going to use the stannous chloride. I told you guys that's my personal favorite. I've got my tin chloride SnCl2 and water. What that's going to give me is my aniline. I'm just going to draw it on this side.
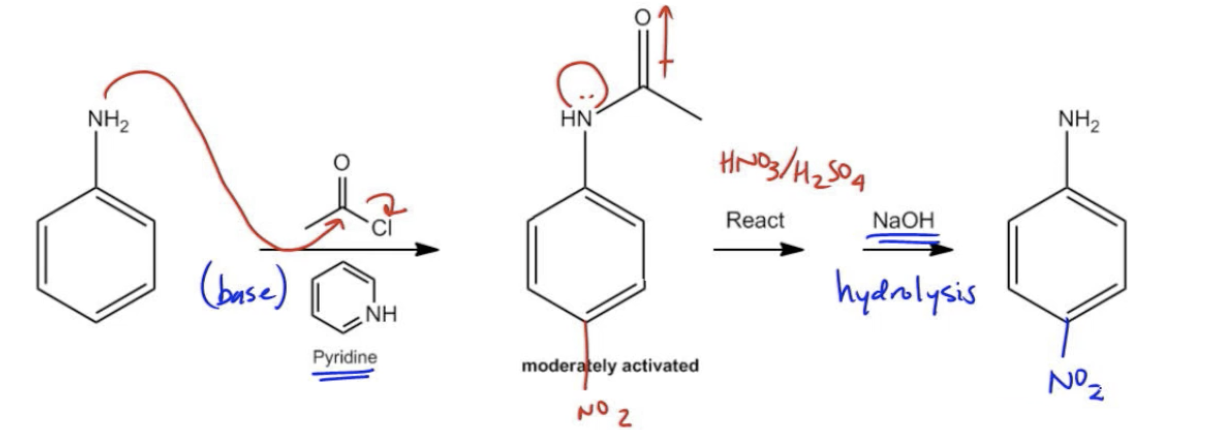
Okay. So now I've got my aniline in place. Can I just do a chlorination? No, I can't. Before this topic, you would have and you would have thought you're doing great. But now because of this topic, we know that you need to acetylate or protect the amino group before we can keep reacting with it. What we're going to do is we're going to add CH3COCl over pyridine. What that's going to give us is now a molecule that look