Back when I taught you guys about functional groups, I told you that alkanes actually don't count as a functional group. Okay? Even though they're super abundant, they're everywhere. And the reason that I said that is because it's true. A functional group implies function, that they actually do something, and alkanes really don't react with much at all. Alkanes just come from underground. You dig them up in petroleum. That's what oil is. It's alkanes, and you can't really react with it much. They're super stable. All you can do is blow them up. Okay? You can put them in your car and combust them, but you can't really react with them a whole lot. So, they seem kind of worthless at first glance, but it turns out that there is one thing that they actually can undergo, and that is that they can undergo a radical reaction. Okay? Because radicals are very high energy. So they're going to be able to react with something that's seemingly unreactive, which is alkanes. So I want to show you guys the mechanism by which they do that. So as I just said, alkanes are the backbone of organic molecules, but they're almost completely unreactive. That's why they last for millions of years underground because they don't react with anything. Okay? But there is one thing that they can do in the presence of radicals, and they can add halogens. Okay? So here I have an unreactive hydrocarbon. Like I said, that's from the dinosaurs. It didn't do anything that whole time. Now, I bring it up to the science lab, and I react it with a radical reaction and, lo and behold, I get a halogen on that alkane. Now, what's cool about that is that now I can do a bunch of other types of reactions to it. This is now called a functionalized hydrocarbon. Why? Because now I have a functional group, an alkyl halide. Once you have an alkyl halide, that's the gateway towards organic synthesis because now, guess what? I can do a bunch of stuff so that I can do substitution reactions, elimination reactions, addition reactions, all kinds of stuff because I first added that halogen. Okay? So what I'm going to show you right now is really the first step of all organic synthesis. Okay?
Free Radical Halogenation - Online Tutor, Practice Problems & Exam Prep
Alkanes are the backbone of organic molecules, yet they are almost completely unreactive.
The only reaction alkanes undergo is radical halogenation, the gateway to the rest of organic synthesis.
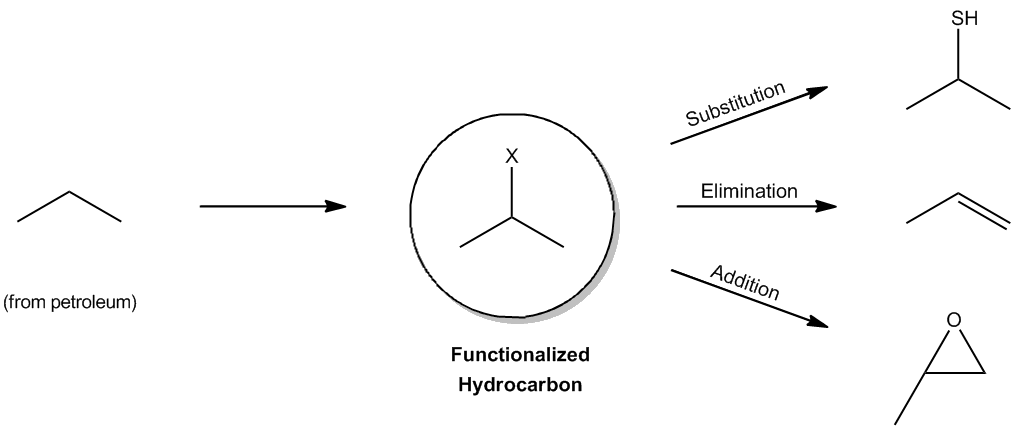
The one reaction that alkanes will actually undergo.
Video transcript
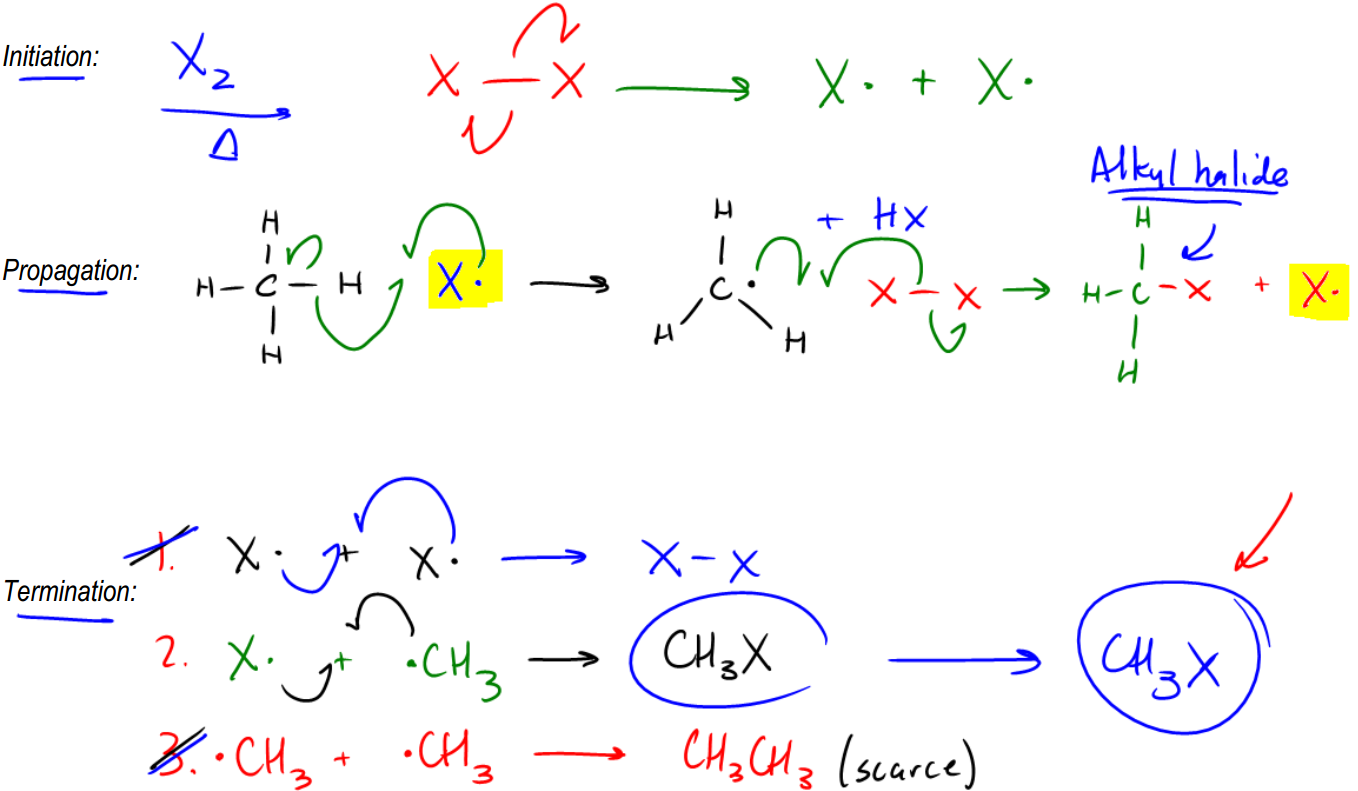
Radical Chain Reaction Mechanism.
Video transcript
So let's just go ahead and talk about it. It turns out that radicals are so high energy that once they react with something, they're going to keep trying to give away that high energy intermediate. Okay? And what winds up happening is that it's like a game of hot potato where no one wants to have the hot potato, so they keep passing it along and it forms what's called a radical chain reaction. Okay? Now the chain reaction, it actually does mean that. It means that once you start it, it actually can't end until it's fully reacted. Until you've fully reacted with all of your alkane. That is useful for us because remember, alkanes aren't that great to begin with, so if we can react them completely, that's a useful reaction as an organic chemist. So let's go ahead and see how this works. Our first step is going to be the initiation step. The initiation step is where I get that first radical because I can't play a game of hot potato without the hot potato itself, so I have to create that first radical. Now, what you notice is that this mechanism is broken down into 3 different steps. And we're actually going to need to write all 3 of these steps. In fact, it's smart that you actually write the words if you do have to draw this mechanism for a test, that you write these three words: Initiation, propagation, and termination. Okay? So let's look at the initiation step. And let's say that we're just using the easiest radical initiator, which is X2. Okay? Let's use X2 over heat. Now, what I taught you guys is that in the initiation step, what we're going to wind up getting is electrons from 2 electrons, 1 on each side, jumping onto each X. So what I'm going to wind up getting is X· + X·. Okay? That's the end of my initiation step. Really all I need for the bare minimum of my initiation step to work, all I need is one radical. In this case, I have 2, so I'm great. Okay? Now that I have that radical in place, that radical is free to react with other molecules. Okay? And it turns out that it reacts really well with alkanes. Now, for the sake of a really simple mechanism, let's just use the simplest alkane possible, which is methane. Methane just being a 1 carbon hydrocarbon. Okay? CH4. So now I've got CH4 and I'm reacting that with X·. Okay? This X· hates itself right now. It's super high energy, super unstable. It's saying, how can I get rid of this hot potato? Can I get rid of it? And then it sees all these electrons in the methane and it's thinking, maybe I can take one of the electrons from one of those carbon-hydrogen bonds. And that's exactly what it does. So it turns out that radicals are going to react with hydrogens and alkanes. And the way we draw these arrows is just so you know, radical reactions are always going to have 3 arrows. So I'm going to draw 1 fishhook into the middle of nowhere. Okay? Then I'm going to draw another fishhook from the CH bond meeting that one. Okay? What that's implying is that now there's going to be a new bond between the H and the X that's going to form from those 2 electrons. So that's looking great, but I still have one electron left over. Notice that the bond between the CH had 2 electrons. So where do you think that last electron goes? It goes onto the C. Okay? It goes onto the carbon backbone. So what that's going to do is it's going to give me a structure that now looks like this: C···HH·. Now notice that I'm drawing the geometry differently because now this would be trigonal planar, right? So you should draw it with like a triangle and that would be + HX. Making sense so far? So notice that the reason this is called a propagation step is that propagation means like I'm reproducing myself. Propagating. And notice that the radical just reproduced itself. Now, it has kind of moved through my medium and now I've got a radical on a new species. Okay? Well, it turns out that your propagation step isn't done yet because you're not done with the propagation step until you fully reproduce yourself. 100%. So what that means is not only do I need to have a radical at the end, I need to have the same exact radical that I started with. So if I start off with an X·, I need to end off with an X·. So what that means is what could I react my C· with to generate that original X· that we had at the beginning? Can you think of anything? It turns out the easiest thing to do is just to react it with another X-X, diatomic halogen. Now, you might be wondering, well, why isn't this already radicals? Because we just did that in the first step. We made it radical. Well, it turns out that not all of the diatomic halogen is going to cleave at the same time. So some of it's going to do the initiation step, but some of it isn't going to be hit by enough light or enough heat to split yet. So what that means is that the one I'm reacting with here hasn't really cleaved yet. Okay? So this is one that's just waiting around for enough energy to finally do that homolytic cleavage. But wait, before the light can even get to it, another radical just did. So instead, we're going to propagate to the X-X. And the way we draw these arrows is once again, 3 arrows. So I'm going to take the radical that always starts it. I'm going to put that one out into the middle of nowhere. Okay? Between the C and then the X. So then I'm going to take one electron from this bond and make it go there. This>
Alkanes will react with diatomic halogens in the presence of heat, light or any other radical initiator.
- Think of the radical as a “hot potato” that the alkanes want to keep passing along!
Explaining the following problem.
Video transcript
Now I want you guys to practice the general mechanism for radical halogenation on your own. And I want you guys to notice that this alkane that I'm reacting with has carbons of different stabilities. Okay? I want you guys just to assume that we're going to react with the most stable carbon in this case. So you're going to have to think back to what I talked about with radical stability to figure out which of those hydrogens to pull off in the radical halogenation. I think you guys can get this, though, so I'm just going to let you guys loose on your own, try to draw all 3 steps, and then I'll give you guys the answer. So go for it.
Show the entire chain reaction mechanism.
Video transcript
We know we need to start with the initiation step here. But even before we get to that, I want to ask you guys which hydrogen did you react the radical with, enoyalkane? Basically, there's actually only one choice that made sense, and you should have reacted it with the H right here. The reason is that this H belongs to the only tertiary carbon on this molecule. Now notice that there are no allylic sites here, so I don't worry about the resonance thing. I just worry about which hopefully this will be a learning experience for you. So now let's go ahead and draw the 3 steps. My first step is going to be initiation. I'll just draw it up here to make more room. Okay. So my initiation step is really easy. We're using XX again, so I'm just going to draw like that and like that. I'm going to do this. And what I'm going to wind up getting is 2 X radicals. Cool?
So now let's go into propagation. Now this is the part where it actually matters which hydrogen I used. And I just showed you guys why we're going to use that hydrogen there. I'm going to redraw my alkane and I'm going to draw the hydrogen sticking off this way this time just to make it easier to pull it off. I'm going to react that with x radical. And what that winds up giving me is 3 arrows. 1 here, 1 here, and 1 there. Okay? So what I wind up getting is a radical that looks like this. Okay? That radical plus HX. Okay? Now, what can that radical react with? Well, it in order to fully propagate and reproduce itself, it's going to have to react with another XX. So I'm going to do this, that, and that. And what that's going to give me is it's going to give me an alkyl halide. Notice that this is now a tertiary alkyl halide because I reacted it at a tertiary position, and I'm going to get that final radical. Cool. So now we're just going to end off with the termination step. And with the termination step, we basically had 3 different possibilities. We had X terminating with X. That would be my first product, and that would give me basically that would give me XX. Then we had another possibility, which was now my alkane radical terminating with a halogen radical. And what that would give me is another equivalent of a tertiary alkyl halide. And then, lastly, I had the third possibility, which would be I have basically 2 R groups colliding with each other. Okay? And that would give me a small amount of this kind of random-looking thing, which is going to be, that and then a single bond. And that single bond is attached to basically 2 more methyls and something like that. Okay? So anyway, I know that's ugly. Okay? But the whole point is that I can change the one. In the end of the day, I'm not going to get a whole lot of 1, I'm not going to get a whole lot of 3, but I am going to get a lot of 2. So my final product would be this guy. And then plus, I would get, obviously, a lot of HX as a byproduct. Okay? Because that's going to pretty much be forming all the time. Alright?
Do you want more practice?
More setsYour Organic Chemistry tutors
- Free-radical chlorination of hexane gives very poor yields of 1-chlorohexane, while cyclohexane can be convert...
- c. How could an industrial plant control the proportions of methane and chlorine to favor production of CCl4? ...
- Peroxides are often added to free-radical reactions as initiators because the oxygen–oxygen bond cleaves homol...
- When exactly 1 mole of methane is mixed with exactly 1 mole of chlorine and light is shone on the mixture, a ...
- Write a mechanism for the light-initiated reaction of cyclohexane with chlorine to give chlorocyclohexane. Lab...
- 3. For each alkane, which monobrominated derivatives could you form in good yield by free-radical bromination?...
- For each alkane, 1. draw all the possible monochlorinated derivatives. c. 2-methylpentane d. 2,2,3,3-tetram...
- The chlorination of pentane gives a mixture of three monochlorinated products. a. Draw their structures.
- Predict the major monohalogenation product(s) of the following reactions. Indicate whether you think the react...
- (••••) When a student attempted a bromination to produce compound A, they generated compound B instead. Ration...
- The radical fluorination of 2-methyl propane resulted in a 14:86 ratio of products. (a) On the basis of this...
- The radical fluorination of 2-methyl propane resulted in a 14:86 ratio of products. (b) From the relative re...
- Show how free-radical halogenation might be used to synthesize the following compounds. In each case, explain...
- Rationalize the fact that reaction A results in an unequal mixture of products, but reaction B yields an equal...
- (••••) Because of the angle strain present in cyclopropanes, they tend to open up in the presence of nearby ra...
- When (1R,3S)-1-tert-butyl-1,3-dimethylcyclopentane is halogenated, one stereoisomer is produced in excess. (a...
- When (1R,3S)-1-tert-butyl-1,3-dimethylcyclopentane is halogenated, one stereoisomer is produced in excess. (b...
- Predict the major products of the following alkane halogenation reactions. [The number of products shown ignor...
- Predict the major products of the following alkane halogenation reactions. [The number of products shown ignor...
- Can you make a 1° bromoalkane like (3-bromopropyl)cyclopentane using alkane halogenation? Why or why not?
- (•) Predict the major product of the following bromination reactions. (b)
- (•) Predict the major product of the following bromination reactions. (c)
- (•••) Suggest a mechanism for the following reactions. (a)
- (•••) For the following reaction, answer questions (a)–(d). (b) Which is the...
- (•••) For the following reaction, answer questions (a)–(d). (d) Given your c...
- Assessments 11.62–11.65 should be answered in order. (•••) A halogenation intended to make compound A formed ...
- Assessments 11.62–11.65 should be answered in order. (•••) A halogenation intended to make compound A formed ...
- In the following reaction, which C―H bond would be most likely to react with a bromine radical?
- Assessments 11.57–11.61 should be answered in order. Each question should be used to help you answer the next....
- Assessments 11.57–11.61 should be answered in order. Each question should be used to help you answer the next....
- Draw the products of the following reactions, including all stereoisomers: d.
- What are the product(s) of each of the following reactions? Disregard stereoisomers. e.
- What are the product(s) of each of the following reactions? Disregard stereoisomers. d.
- What are the product(s) of each of the following reactions? Disregard stereoisomers. c.
- How many alkyl halides are obtained from monochlorination of the alkanes in Problem 4 if stereoisomers are inc...
- How many alkyl halides are obtained from monochlorination of the alkanes in Problem 4 if stereoisomers are inc...
- How many alkyl halides are obtained from monochlorination of the alkanes in Problem 4 if stereoisomers are inc...
- How many alkyl halides are obtained from monochlorination of the alkanes in Problem 4 if stereoisomers are inc...
- How many alkyl halides are obtained from monochlorination of the alkanes in Problem 4 if stereoisomers are inc...
- How many alkyl halides are obtained from monochlorination of the alkanes in Problem 4 if stereoisomers are inc...
- How many alkyl chlorides are obtained from monochlorination of the following alkanes? Disregard stereoisomers....
- How many alkyl chlorides are obtained from monochlorination of the following alkanes? Disregard stereoisomers....
- How many alkyl chlorides are obtained from monochlorination of the following alkanes? Disregard stereoisomers....
- How many alkyl chlorides are obtained from monochlorination of the following alkanes? Disregard stereoisomers....
- How many alkyl chlorides are obtained from monochlorination of the following alkanes? Disregard stereoisomers....
- How many alkyl chlorides are obtained from monochlorination of the following alkanes? Disregard stereoisomers....
- What are the product(s) of each of the following reactions? Disregard stereoisomers. f.
- What are the answers to Problem 29 when the same compounds are treated with Br2 at 125 °C? c.
- What is the major product obtained from treating an excess of each of the following compounds with Cl2 in the ...
- What alkyl halide will be obtained in greatest yield? Ignore stereoisomers. a.
- What alkyl halide will be obtained in greatest yield? Ignore stereoisomers. b.
- a. What hydrocarbon with molecular formula C4H10 forms only two monochlorinated products? Both products are ac...
- b. What hydrocarbon with the same molecular formula as in part a forms three monochlorinated products? One is ...
- Explain why iodine (I2) does not react with ethane, even though I2 is more easily cleaved homolytically than t...
- Write the initiation, propagation, and termination steps for the monochlorination of cyclohexane.
- Provide an arrow-pushing mechanism for the following alkane bromination. [Don't forget to use fishhook arrows ...
- (•••) If a small amount of a moderately nonpolar poisonous compound was added to a pond, why would it be safe...
- Predict the major monohalogenation product(s) of the following reactions. Indicate whether you think the react...
- (••) Predict the product(s) of the following halogenation reactions. Only one equivalent of the halogen is us...
- (••) Predict the product(s) of the following halogenation reactions. Only one equivalent of the halogen is us...
- We have studied the following reactions in previous chapters. For each, (i) indicate which reaction sheets the...
- Provide a mechanism for the chlorination of cyclohexane. Be sure to include initiation, propagation, and three...
- (•••) For the following reaction, answer questions (a)–(d).<IMAGE>(a) Give an arrow-pushing mechanism, i...
- Predict the major products of the following alkane halogenation reactions. [The number of products shown ignor...
- Assessments 11.57–11.61 should be answered in order. Each question should be used to help you answer the next....
- Assessments 11.57–11.61 should be answered in order. Each question should be used to help you answer the next....
- a. What alkane, with molecular formula C5H12, forms only one monochlorinated product when it is heated with Cl...
- Explain why the rate of bromination of methane decreases if HBr is added to the reaction mixture.
- How many alkyl halides are obtained from monochlorination of the alkanes in Problem 4 if stereoisomers are inc...
- c. How many monochlorination products would be obtained if all stereoisomers are included?
- What is the major product obtained from treating an excess of each of the following compounds with Cl2 in the ...
- In the presence of an acid catalyst, acetaldehyde forms a trimer known as paraldehyde. Because it induces slee...
- a. Propose a mechanism for the following reaction:<IMAGE>
- How many alkyl halides are obtained from monochlorination of the alkanes in Problem 4 if stereoisomers are inc...
- a. How many monochlorination products can be obtained from the radical chlorination of methylcyclohexane? Disr...
- Write the steps for formation of tetrachloromethane (CCl4) from the reaction of methane with Cl2 + hv.
- A possible alternative mechanism to that shown in Problem 47 for the monochlorination of methane involves the ...
- Show how you would prepare cyclopentene from each compound.c. cyclopentane (not by dehydrogenation)
- Each of the following proposed mechanisms for the free-radical chlorination of methane is wrong.Explain how th...
- a. Propose a mechanism for the free-radical chlorination of ethane,CH3—CH3 + CL2 hv —> CH3—CH2Cl + HCl
- a. Draw the structure of the transition state for the second propagation step in the chlorination of methane.⋅...
- Using the BDEs in [TABLE 4-2] <IMAGE> (page 167), c. Suggest two reasons why iodine does not react well...
- Alkoxy radicals (R—O•) are generally more stable than alkyl (R⋅)radicals. 1. Write an equation showing an alky...
- Using cyclohexane as one of your starting materials, show how you would synthesize the following compounds. (a...
- When exactly 1 mole of methane is mixed with exactly 1 mole of chlorine and light is shone on the mixture, a ...
- a. Write the propagation steps leading to the formation of dichloromethane (CH2Cl2) from chloromethane.
- Each of the following proposed mechanisms for the free-radical chlorination of methane is wrong. Explain how t...
- a. Propose a mechanism for the free-radical chlorination of ethane, CH3—CH3 + CL2 hv —> CH3—CH2Cl + HCl b. ...
- For each alkane, 1. draw all the possible monochlorinated derivatives.a. Cyclopentane b. Methylcyclopentane
- 2. For each alkane, determine whether free-radical chlorination would be a good way to make any of these monoc...
- b. Explain why free-radical halogenation usually gives mixtures of products.
- c. How many dibrominated products could each of the compounds form if stereoisomers are included?
- a. The following compounds have the same molecular formula as benzene. How many monobrominated products could ...
- Free-radical bromination of the following compound introduces bromine primarily at the benzylic position next ...
- (•••) FROM THE LITERATURE The following transformation was found to occur in areas with large NO₂ emissions. S...
- The human body can excrete drugs and other exogenous molecules by converting them into polar, water-soluble co...
- b. For each reaction, show which stereoisomers are obtained1. NBS/∆/peroxide2. Br2/CH2Cl2
- (••••) One danger associated with storing ether solvents is their tendency to form explosive peroxides when ex...
- a. Which ether is most apt to form a peroxide?
- b. Which ether is least apt to form a peroxide?
- Write an equation for the reaction of vitamin E with an oxidizing radical (RO·) to give ROH and a less reacti...
- A student adds NBS to a solution of 1-methylcyclohexene and irradiates the mixture with a sunlamp until all th...
- For each compound, predict the major product of free-radical bromination. Remember that bromination is highl...
- Using the bond-dissociation energies in Table 5.6, (a) predict whether or not an iodine radical would be se...
- a. What is the major product of the reaction in Problem 7 when the alkane reacts with Cl2 instead of with Br2?...
- Radical addition to alkenes is not effective for the synthesis of iodo- and chloroalkanes. Using your knowledg...
- The benzene ring alters the reactivity of a neighboring group in the benzylic position much as a double bond ...
- Show how the following compounds could be prepared from 2-methylpropane:a. 2-bromo-2-methylpropane

