In this set of videos, I want to talk about predicting acidity for phenols. Phenols are alcohols but they're substantially more acidic than a regular alcohol due to the resonance effect. Remember that you had these effects in your acid-base chapter way back in the day that told you when something was going to be a better acid. It said that if you're able to stabilize the conjugate base, then your acid will be more acidic. Think about phenol, guys. Phenol, after it gives up its proton, becomes phenoxide. Phenoxide, it's a negative charge. Negative charges aren't that happy. But phenoxide can resonate. Notice that it can make resonance structures inside the ring. We'll be able to resonate the negative charge to here, here, and here. What that means is that normally the pKa of an alcohol is about 16, the same as water pretty much. But the pKa of phenol is closer to 10. That's because of this resonance-stabilized conjugate base that you can make. It's more stable, and therefore, the phenol is going to be more willing to give up its proton. It turns out that we need to also understand how electron-withdrawing groups and electron-donating groups play into this situation as well. In this example, when I have D, that just means it stands for ED group, an electron-donating group. When I have W, that's an electron-withdrawing group. In general, we can say that if you're pushing electrons into the ring, do you think that's going to make it more acidic or less acidic if you're a donating group? That's going to make it less acidic because of the fact that you're destabilizing the conjugate base. The conjugate base already has a full negative charge. Do you think it wants more electrons being jammed up into that benzene? No. Whereas, what do you think about electron-withdrawing groups? Absolutely. That's going to make it more acidic. The more electron-withdrawing groups we have, the better. That's going to pull more electron density out of the ring and it's going to stabilize that conjugate base. Got it? Really quick, let's just do a quick example already. Go ahead and look at these four phenyls and tell me which one you think is going to be the most acidic phenyl.
Phenol Acidity - Online Tutor, Practice Problems & Exam Prep
Donating vs Withdrawing Groups
Video transcript
Identify the most acidic phenol
O,P-positions vs. Meta-Positions
Video transcript
It turns out that not all positions are created equal. It turns out that some positions are going to have more effect on acidity than others. In fact, the meta position is going to have a much lesser effect on acidity than the ortho and para positions, meaning that whatever type of group is on there, it just matters less if it's on the meta and it matters more if it's on an ortho or para. Why is that? Think about the resonance structures that occur when you make a phenoxide. Remember that I told you guys where would that negative charge resonate to? It would resonate to the top, to the side, and to the side. Notice what are those positions called respective to the O. Those are the ortho para positions. That means the negative charge will rest directly on ortho para positions and it will never rest on a meta position. With that logic, what that means is that we know that a donating group is going to make it less acidic because it's going to destabilize the negative. We know that a withdrawing group is going to make it more acidic. But if those same exact groups are in the meta position, so these are meta and these are ortho. I'm looking at orthos versus meta. If I have a donating group in the meta position, it is going to be a little less acidic but only slightly less acidic than normal. Why? Because this donating effect doesn't really matter that much because it's on the meta position. The negative charge never goes there. Same thing goes with the withdrawing group. With withdrawing group, you think this is great. It's going to make it really acidic. But it's only slightly more acidic if you rest on the meta position. Why? Because once again, the meta position doesn't really matter because the negative charge never actually sits on it. In terms of acidity, that brings us to the following acronym. If you're looking for an acidic phenol, what you're going to be looking for is a WAP. What a WAP stands for is a withdrawing group in the ortho and para positions. You don't care about the meta positions. The meta positions aren't helpful for us. We're looking mostly at those having withdrawing groups in the ortho and para positions. That being said, why don't we take a stab at this question and tell me which one you think is going to be the most acidic phenol.
Identify the most acidic phenol
Video transcript
Notice that my groups are identical for all 4 phenols. The only difference is the positions that they're in. What I have for the first one is that I have an ortho, a meta, and a para. These are all withdrawing groups, very strong withdrawing groups. What I have for the second one is an ortho, a meta, and a meta. What I have for the third one is an ortho, a para, and a meta. Finally, what I have for the fourth one is an ortho, para, and ortho. Which one is going to win? Which one is going to have the most stabilizing effects on that phenoxide? It's obviously going to be D because D is the WAP. D has withdrawing groups on the ortho and para. Now, does that mean the other ones aren't acidic? No. They're very acidic. They're doing great. But this one's a little bit better because all of the groups happen to be in those ortho and para positions. It's going to be able to stabilize the negative charge in every position that it goes with the withdrawing group. Make sense? Cool guys. Let's turn the page.
Identify the most acidic phenol
Video transcript
Which one was the most acidic phenol in this case? Actually, I'm just going to tell you right now. The answer was A. Did you pick A? Yikes. I'm sensing that a lot of you did not pick A. But that's because maybe you forgot about withdrawing and donating groups. What kind of group is NMe2? This is going to be an amine derivative or an aniline derivative because you've got a lone pair right there. That means that this is a strong donating group, electron donating group. What did we learn about electron donating groups? Do they make a molecule or phenol more acidic or less acidic? They make it less acidic. We found out that if we're going to have a withdrawing group, then we want it to be in the ortho position. Destabilize your anion. Destabilize your anion. Then you want it to be in the meta position because the meta position is going to have the smallest effect. When you don't have any withdrawing groups, then you go to donating groups and you look for them in the meta position. In this case, what I have is 2 donating groups. That one's immediately going to lose because I don't want 2 of them. They're already bad as bad as they are. I don't want 2 things that are destabilizing my phenol. That's terrible. Then I have ortho. Then I have a donating group that's ortho versus a donating group that's meta. The donating group that's meta is going to win because the donating group that's ortho is going to be significantly destabilized when my negative charge goes there. But my donating group that's meta will be fine because the negative charge never actually hits that carbon, so it's not going to be as destabilized, and it's going to continue to remain acidic. We have to think kind of backwards here. Since we weren't given any withdrawing groups, you have to go with the donating group that has the lowest effect on the phenol. The answer is A. Here's another one. It's going to be tricky. Try your hardest and then I'll tell you the answer.
Identify the most acidic phenol
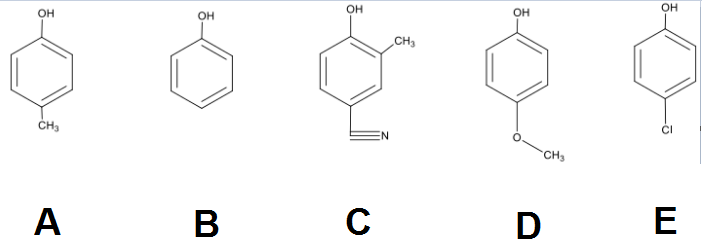
Rank the following phenols in order of increasing acidity.
Do you want more practice?
More setsYour Organic Chemistry tutors
- (•••) Benzoic acid and phenol are insoluble in water. When sodium bicarbonate is added to the water, benzoic ...
- The following compounds are only slightly soluble in water, but one of them is very soluble in a dilute aqueou...
- The following compounds are only slightly soluble in water, but one of them is very soluble in a dilute aqueou...
- Predict the products formed when m-cresol (m-methylphenol) reacts with (b) acetyl chloride,
- Predict the major products of the following reactions.(b) phenol + tert-butyl chloride + AlCl3
- (••) The dye alizarin normally forms an orange solution when dissolved. However, if KOEt is added to the solut...
- (•••) Rationalize the fact that 1,4-dihydroxybenzene melts at a significantly higher temperature than 1,2-diyd...
- (••) Sulpiride is an antipsychotic. Provide the reagents (a) and (b) used to complete the synthesis.<IMAGE&...
- (•••) Phenol is synthesized from cumene via the scheme shown. Suggest the mechanism for each step of the react...
- Though the nitro group is electron-withdrawing by resonance, when in the meta position, it doesn’t communicate...
- e. Which has a greater Ka: cyclohexanol or phenol? f. Which is a stronger acid: cyclohexanol or phenol?
- Rank the amines in each set in order of increasing basicity.(c) <IMAGE>

