On this page, we're going to talk about a reaction called enamine alkylation and acylation. Just to catch you up, in the ketones and aldehydes section of your textbook, there's a reaction that ketones and aldehydes can undergo with amines that forms imines and enamines. These are functional groups that form from the addition of neutral amines to a ketone or aldehyde. Specifically, the one that we care about for this page is secondary amines because as you might remember, or maybe you haven't gotten there yet, and it's fine, or you just forgot, secondary amines are going to react with ketones to specifically give enamines. An enamine has a part amine at the top and an alkene at the bottom, hence the name. Enamines are really important for us for one reason alone, which is that enamines have a nucleophilic alpha carbon. If you think about it, this was the alpha carbon to begin with. This is still the alpha carbon. Enamines are able to use that alpha carbon to do nucleophilic attacks. Enamines have the ability to alkylate or acylate via the formation of an iminium salt. What essentially happens is that you have your enamine and you have an electrophile. Let's just say that it's an alkyl halide to keep it easy. What happens is that the lone pair from the nitrogen can come down to make a double bond. You make that bond. You break a bond. You take this double bond and you attack the alkyl group with it because it's electrophilic. What you end up getting is the formation of an iminium salt. Really important, iminium salt because now that nitrogen has a positive charge. But more importantly, we just alkylated the alpha carbon. Then we can use dilute acid to hydrolyze the iminium salt all the way to a ketone. If you're wondering how that happens, this happens through what we call an acid workup. This is a very important reaction in organic chemistry 2. We need to know how to hydrolyze nitrogen compounds into oxygen-containing compounds. This is a multiple-step reaction. But if you're interested in looking it up, you can look it up in the basically the imine topic. When we discuss imines in the carbonyls section, Ketones and Aldehydes, we're going to talk about the forward reaction of this, how to go from a ketone to an imine. The reverse of that imine reaction is what we call an imine hydrolysis to get back to the ketone. I'm not going to go through that whole mechanism now because I already have in prior videos. But just letting you know that we basically use acid to turn an imine derivative into a carbonyl. But now it's not just a carbonyl. It's alpha-substituted. What I want you guys to do is go through these two examples. Try to solve them yourself. Do the first one first, obviously, and then I'll solve the first one, and then we'll do the second one together. Go ahead and do the first one.
Enamine Alkylation and Acylation - Online Tutor, Practice Problems & Exam Prep
General Reaction
Video transcript
Predict the Products
Video transcript
All right guys, so as you'll notice in this question, it's not asking for a full mechanism. It would take over a page to draw this whole mechanism from scratch. We're just going to predict the products like it says. The first thing, the first two reagents work together. These are going to be an acid catalyzed enamine reaction. If you're wondering how to draw the product of an enamine, I always like to rely on the general formula. What I would do is I would draw that an enamine is an N with 2 bonds at the top, a bond coming down and then a double bond coming out from one of its sides. Then I just have to determine what are the R groups that I substitute for everything. The N is actually supposed to be attached to a square. I'm going to go ahead and complete the square at the top. The bottom was actually coming from the carbonyl which was acetone, so it's already drawn perfectly. Now it's time for the reaction, for the
Predict the Products
Provide the major product after each step for the following reaction.
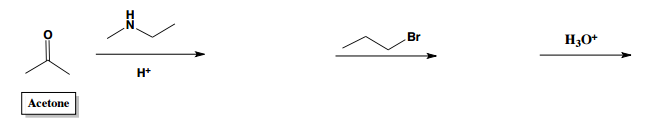
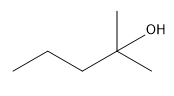
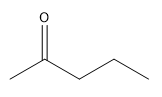


Which of the following can NOT be formed through the stork enamine reaction with 2-butanone?
Do you want more practice?
More setsYour Organic Chemistry tutors
- Describe how the following compounds could be prepared from cyclohexanone using an enamine intermediate: b.
- Write equations showing the expected products of the following enamine alkylation and acylation reactions. Th...
- Without looking back, propose a mechanism for the hydrolysis of this iminium salt to the alkylated ketone. The...
- Write equations showing the expected products of the following enamine alkylation and acylation reactions. The...
- Describe how the following compounds could be prepared from cyclohexanone using an enamine intermediate:a. <...
- What reagents should be used to prepare the following compounds?c. <IMAGE>
- Show how you would accomplish each conversion using an enamine synthesis with pyrrolidine as the secondary ami...
- Predict the products of the following reactions. (e)

