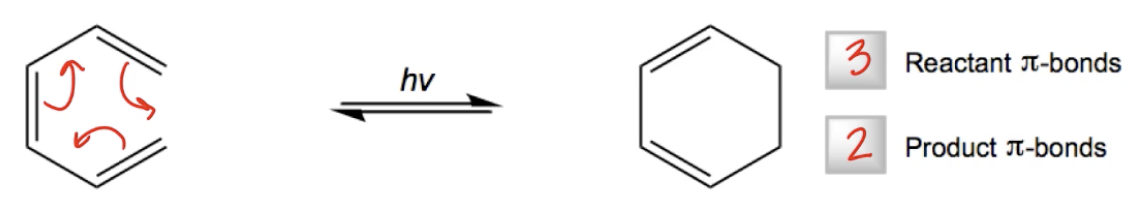
Hey, everyone. In this video, we're going to discuss a type of pericyclic reaction called a photochemical electrocyclic reaction. So photochemical electrocyclic reactions are simply going to be intramolecular pericyclic reactions in which 1 pi bond is destroyed after a light-activated cyclic mechanism. I know that's a mouthful, but you guys should already be really comfortable with all those key terms. It's intramolecular because all electrocyclic reactions are intramolecular. It destroys 1 pi bond because all electrocyclic reactions destroy 1 pi bond, and it's light-activated because we're using photochemical energy. So here's an example, we have a molecule that is reacting with itself in the presence of light to form a new ring and we are changing 1 pi bond in the process. We start off with 3 and we end up with 2 and this mechanism would be the same exact for the thermal, conversion, electrocyclic reaction because nothing has changed. All that's going to happen is that you're going to form a new sigma bond and then go through the rest of your concerted mechanism. And basically, in the meantime, you exchange 1 sigma bond for 1 pi bond. Cool? Awesome. So by the way, just want to throw this out there, every single conjugated polyene is capable of doing this. So it's not unique to specific types. Any polyene can do this, but the stereochemistry does depend on Frontier Molecular Orbital Theory. So what we're going to be doing, we're not going to focus too much on the general mechanism because that's the easy part. We know we're going to form a ring. We're going to focus more on the idea of HOMO and LUMO frontier orbitals, so that we can figure out what the stereochemistry of the product will be. Okay. Now something that is unique to a photochemical electrocyclic reaction is that light is going to be involved in exciting ground state electrons and kicking them up one energy level. So it's going to take those electrons in their ground state and it's going to move them to a higher energy state. So usually that means we're going to go from a bonding Psi to an anti-bonding Psi, and that means that your HOMO and your LUMO orbitals are going to change. And since the stereochemistry of an electrocyclic reaction is dependent on understanding the HOMO orbital, that means that we need to take light into account because it's going to change the identity of the homo molecular orbital. Okay? So let's go ahead and look here at just basically a diene, which is a very simple, example. And, before we even start, why don't we fill in what the molecular orbitals would look like for a diene. Just remember I'm just going to go through this very quickly because this is not the point of this video, but very quickly the first one doesn't change, the last one always changes and my nodes keep increasing. So this would be 1 node and this would be 2 nodes and this would be 3 nodes. Cool. Awesome. So what we know about a typical diene is that 4 electrons will fill orbitals Psi 1 and Psi 2, making my HOMO Psi 2 usually and my LUMO Psi 3, right. But after I react with light, what's going to happen is that one of these electrons is going to get kicked up to a higher energy state, meaning that now my molecular orbital diagram will look like this. Meaning that my HOMO and my LUMO orbitals have changed. Now this is my new HOMO, Psi 3 and this is my new LUMO, Psi 4. Cool? Now guys, actually LUMO is going to be completely irrelevant for this specific reaction because the electrocyclic reaction is intramolecular and only involves the HOMO of the molecule, but it's just interesting to see how light has now changed the identity of my HOMO orbital. So now when I go ahead and consider the stereochemistry of this molecule, I'm going to have to draw my orbital differently because light was included. So in the next example, in the next video, I'm going to go through an example showing how to draw the stereochemistry from scratch of an electrocyclic reaction that's using light.
Photochemical Electrocyclic Reactions - Online Tutor, Practice Problems & Exam Prep
Photochemical Electrocyclic reactions are pericyclic reactions in which 1 pi bond is destroyed after a light-catalyzed cyclic mechanism.
MO Theory of Photochemical Electrocyclics
Video transcript
Predicting Electrocyclic Products
Video transcript
Predict the product of the following electrocyclic reaction and label the reaction as either conrotatory or disrotatory. So, guys, where should we start? In the same exact place that we started off, with the last with the thermal electrocyclic reaction. Let's see if my paste still works. Oh hell yeah, I got lucky that time. So my paste is still working so I didn't have to draw all my orbitals from scratch. If you haven't drawn these yet, feel free to pause the video until you're ready. So we have to fill in our orbitals, and I'm going to rush through it because you guys should be pros at filling in the orbitals for a diene at this point. Orbitals don't change, these orbitals keep flip-flopping, and then I fill in my nodes. Cool, awesome. And we know that we would have our sides, Ψ₁, Ψ₂, Ψ₃, and Ψ₄. And we know that we would have 2 electrons in each of Ψ₁ and Ψ₂. Now what's going to happen in the presence of light is that we're going to kick it up a notch, literally, and we're going to take one of these electrons and excite it to side 3, meaning that my new HOMO is going to be And what this means is that now, when I draw my three-dimensional representation of this molecule, I have to draw and not So let's go ahead and check it out. Let's try to draw a three-dimensional representation. So it's going to look something like this, like this, and then like this. Cool! Let's draw our orbitals in. 1, 2, 3, and 4. Let's also draw our substituents, which should be facing in on the same plane. Cool! In this case, because my substituents are in, they are in on the same plane. Another way to think about this is that you could draw them into the page and out of the page. So what that means is that this one should be coming out of the page, right, and this one should be going into the page. Cool? Awesome! So now let's fill in our orbitals, and since this is side 3, that means that this one should be shaded at the bottom, these 2 should be shaded at the top, and then this one should also be shaded at the bottom. So when we go to make our new Sigma bond, how do we want to rotate this guy? So what we want to do, what we need to do, is we're going to need to rotate disrotatory, right, so that we can get 2 of the same ends overlapping. So that means if this one goes clockwise, this one must go counterclockwise, right. And what that's going to do is it's going to form a new thing that looks like this. And I didn't draw this last time, but I'll draw it here really quick. These orbitals will look like this now, where now the dark parts are here and here and the light parts are in the middle. I have a double bond here. I can ignore the other orbitals because they're just sitting there. And then where are my substituents? Well, let's look at the first one. Okay, the first one is going into the page, and now it is rotating down, so that means that it should be facing down. Cool? Awesome! Now notice that the one on the top was coming out of the page, right, it's coming out of the page and what I notice is that it's also rotating down, right. So it's coming out of the page and it's rotating down as well. So what that means is that it should face down here. And What that means is that my final product should look like this. Double bond, dash, and dash. Isn't that cool? And that would be our product. Now the question is, are there 1 product or are there 2 products? Should I also draw the product of the other direction if we would had gone one was counterclockwise and one was clockwise with them both on wedges? The answer is no. There's no enantiomer because this is a meso compound. So there's only one product; you should not draw the other one with the wedges because it's the same exact molecule and your professor could technically give you points off because you drew the same molecule twice. Make your professor think that you don't know what's going on, and you don't want to do that. Great, and now we just have to label this rotation. I'll label it on the equilibrium arrows here. This is what we would consider to be dis because they went in different directions. Isn't that cool? Awesome, guys! So now, at this point, you should feel very comfortable with drawing the products of both a thermal and a photochemical electrocyclic reaction. Let's go ahead and move on to the next video.
Do you want more practice?
More setsYour Organic Chemistry tutors
- c. Under photochemical conditions, will ring closure be conrotatory or disrotatory? d. Will the product have t...
- Explain why the hydrogen and the methyl substituent are trans to one another after photochemical ring closure ...
- Draw the product of each of the following reactions:c.<IMAGE>d.<IMAGE>
- Draw the product formed when each of the following compounds undergoes an electrocyclic reactionb. under photo...
- Chorismate mutase is an enzyme that promotes a pericyclic reaction by forcing the substrate to assume the conf...
- Predict the product of the following electrocyclic reactions.(c) <IMAGE>
- Draw the product of each of the following reactions:d. <IMAGE>

