Hey everyone. In this video, I want to introduce a really important concept in organic chemistry called frontier molecular orbital theory. Let's take a look. Frontier orbital interactions are actually the driving force behind many reactions in organic chemistry. So this information that we're going to learn now is actually going to be foundational to understand reactions later. So it's really important that we know what this FMOT theory is, okay. And, the most important thing that we need to know about frontier molecular orbitals is how to find HOMO and LUMO. So you might have heard about HOMO and LUMO before, but in this lesson we really need to fully understand it. So the HOMO is defined as the highest occupied molecular orbital. So out of all your molecular orbitals, it's the one that has the highest energy that still has electrons in it. Okay. Then there's the LUMO. The LUMO is the lowest unoccupied molecular orbital. So once again, out of all of your molecular orbitals, it's the one that has the lowest energy while still having no electrons in it. Okay. So if we were to look at ethene as an example, so ethene is a very simple molecule, so it's easy to understand HOMO and LUMO. So, what we would have is we have 2 atomic orbitals right? And those 2 atomic orbitals form 2 molecular orbitals, we have the bonding orbital, we have the antibonding orbital, and we know that according to the principles of electron configuration, these 2 electrons should only fill the bottom pi one orbital. It's called pi 1, not psi 1 because it's just a double bond, so you just call it pi 1. And that means pi 2 star, which is our antibonding orbital, is unfilled. Okay. So what that means is that our HOMO orbital is pi 1. So we would say that the HOMO contains 2 electrons, it's pi 1 and that means that my LUMO orbital is pi 2 star and it has 0 electrons. That's it. So HOMO is the highest one that still has electrons, which in this case it's the only one because it's the only one that exists, and LUMO has no electrons in it and it's the lowest energy state. But since there are only 2, it just happens to be the only one that doesn't have electrons. As your molecules get bigger and the number of molecular orbitals increases, finding HOMO and LUMO will be on that basically the cusp of where the electrons end and where the unfilled orbitals begin. So let's go ahead and try out a practice problem.
16. Conjugated Systems
HOMO LUMO
16. Conjugated Systems
HOMO LUMO - Online Tutor, Practice Problems & Exam Prep
Frontier orbital interations are the driving force behind many Organic Chemistry recations. Let's find out why.
1
concept
HOMO vs. LUMO
Video duration:
2mPlay a video:
Video transcript
- HOMO = Highest Occupied Molecular Orbital
- LUMO = Lowest Unoccupied Molecular Orbital
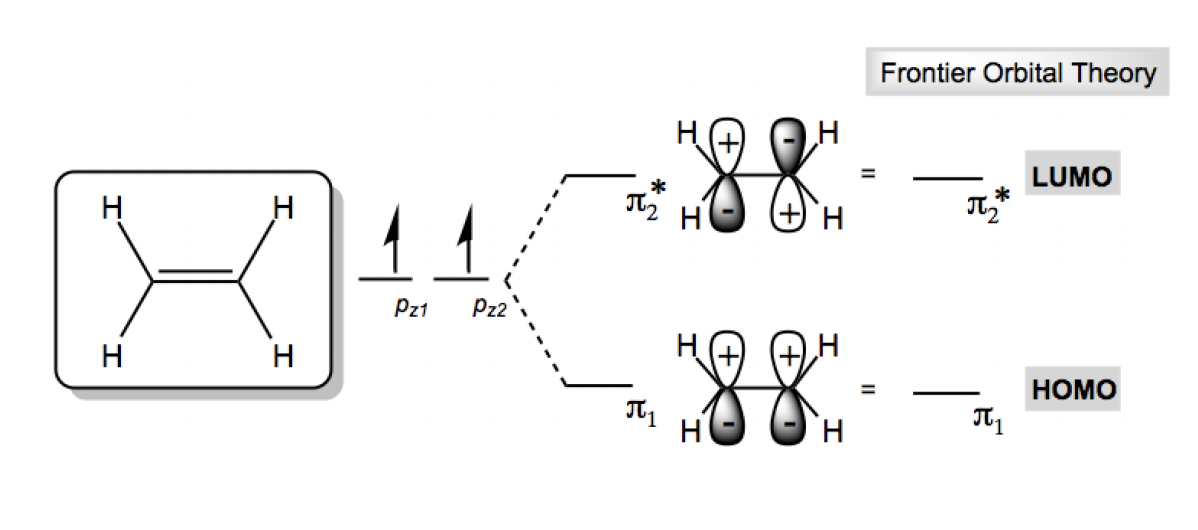
2
Problem
ProblemConsider the MO's of allyl anion. Which of the following are HOMO and LUMO?
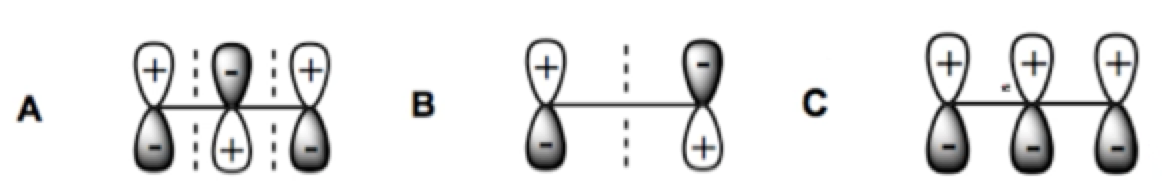
A
HOMO = B, LUMO = C
B
HOMO = B, LUMO = A
C
HOMO = C, LUMO = A
D
HOMO = A, LUMO = C
E
HOMO = C, LUMO = B
Do you want more practice?
More setsYour Organic Chemistry tutors
Additional resources for HOMO LUMO
PRACTICE PROBLEMS AND ACTIVITIES (9)
- Identify the HOMO and LUMO of the allylic cation and the allylic anion shown in Figure 21.22.
- Draw the molecular orbital picture for (a) ethene. Label the HOMO and LUMO of each.<IMAGE>
- Predict the lmax of the following compound:
- Match four of the following UV absorption maxima (λmax) with the corresponding compounds: (1) 232 nm; (2) 256...
- How can you use UV spectroscopy to distinguish between the compounds in each of the following pairs? a.
- Rank the following compounds in order of increasing lmax:
- Rank each set of compounds in order of decreasing lmax: b.
- Rank each set of compounds in order of decreasing lmax: a.
- Which of the following are correct? Correct any false statements c. A conjugated diene with an odd number of d...

