Are you guys ready to have some serious fun? Good, because this topic is going to get nuts. I'm serious. Anyway, we've learned from prior topics that 1,5-dicarbonyls have the ability to undergo intramolecular self-condensation and form 6-membered enones. Remember, we drew these before. What's different now is that we've also learned a reaction that makes 1,5-dicarbonyl compounds, and that's a Michael reaction. When the 1,5-dicarbonyl is generated from a Michael reaction, that means that you had an enone and an enolate come together to make a 1,5-dicarbonyl. When that molecule cyclizes, that's called a Robinson annulation. Essentially, this is an aldol times three. How is that? Well, because think about it. You had to use one aldol to make the original enone, the alpha, beta unsaturated, that's one aldol. Then use the second aldol, the enolate, to attack it via conjugate addition. When you did the conjugate addition, now you made the 1,5-dicarbonyl. But what we're saying now is that the 1,5-dicarbonyl is going to cyclize on itself and make a new 6-membered enone. That's the third aldol and that's all enolate mediated. I know. It's too much fun, right? You're not allowed to have this much fun in organic chemistry. Anyway, here's an example of a Michael product because I've got a 1,5-dicarbonyl. Again, I would be faced with the choice: where do I put the enolate? Do I put it in the blue or in the red position? You're going to put it in the position that's going to give you the 6-membered ring. If I use the red one to attack this carbonyl, do I get a 6-membered ring? I get 4: 1, 2, 3, 4. However, if I use the blue one, 1, 2, 3, 4, 5. Yes, I get what I'm looking for, guys. This is called the Robinson annulation. What I'm going to do first is I'm going to help you guys draw the product and then we'll do one from scratch. For this, this molecule is really easy to draw. Once you have everything numbered out, you can draw your 6-membered ring. I'm going to continue to keep those positions the way that I had them where I have 1, 2, 3, 4, 5, 6. Now I just have to figure out what am I missing on each of these atoms. Position 2 is going to be a ketone. Position 6 is going to be an alcohol and it's also going to have a methyl group. That's the product. But we know that these like to dehydrate. I guess I shouldn't have drawn it in the box. My bad. But the final product of this would be a methyl group here, a double bond here, and that is your cyclic enone. We've done this before. But now, annulation because it started off with a 1,5-dicarbonyl that was made through a Michael reaction. It is similar to what we've already done but just connected to conjugate addition. What I want you guys to do for this product is notice that I'm starting off from the Michael reaction. In this one, I'm starting from Michael because what I have is an enone and what is going to be an enolate. I want you guys to react those together, get your Michael product, and then do the Robinson annulation for that. Try your hardest, and then I will come in and save the day eventually.
Robinson Annulation - Online Tutor, Practice Problems & Exam Prep
1,5-dicarbonyl compounds generated from the Michael Reaction (enone + enolate) have the ability to undergo intramolecular self-condensation into 6-membered enones. Are you impressed yet?
Robinson Annulation
Video transcript
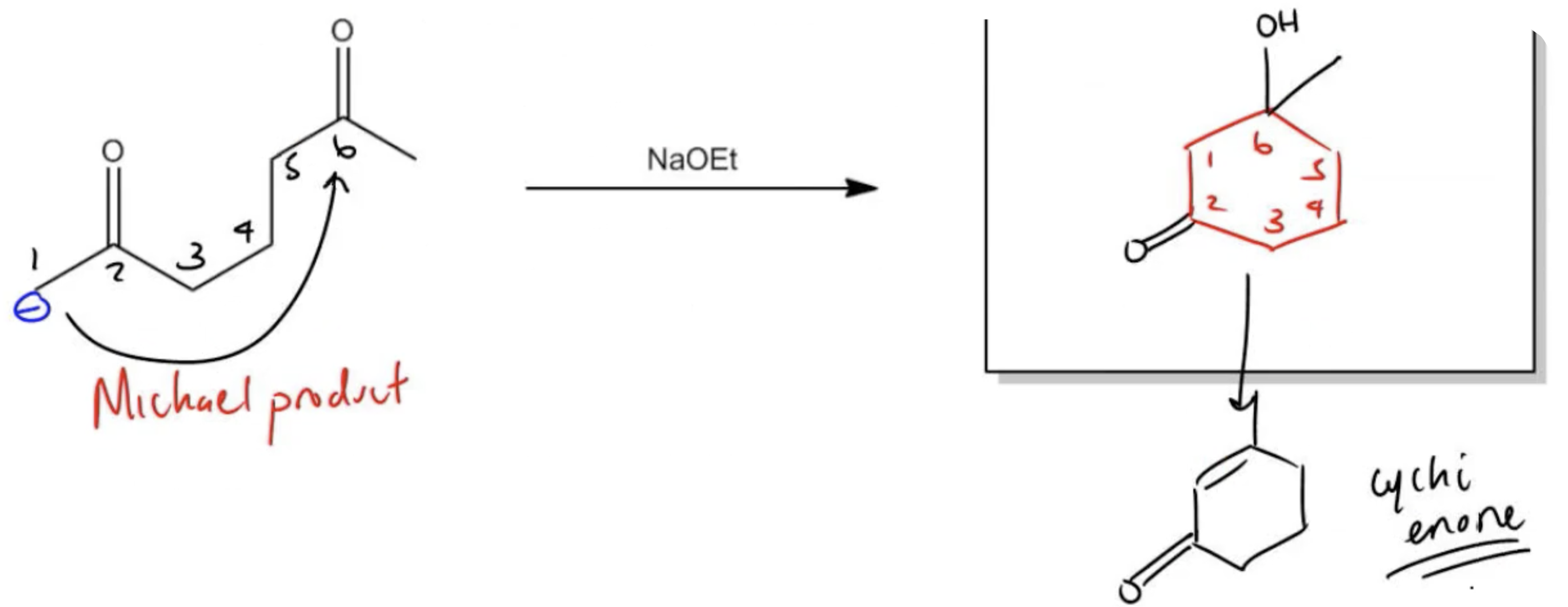
Predict the Major Product
Video transcript
Let's start off with the Michael product. That part's pretty easy to draw, guys, because I told you that for Michael reactions, I always like to draw the enol in the same way it was before and then just put the enolate on top. I would just put my enolate on the top like this. That's not bad because I know that my enolate is going to form there. Fair? Cool. Now I have to figure out where to form an enolate so that it can cyclize into a 6-member ring. I've got, I guess, 4 possibilities. I've got my black, I've got my red, I've got my blue, and I've got my green. Notice I've got a bunch of different possibilities here. What we're wondering is which one is going to make the best 6-member ring. There are 2 that I can count out immediately and that's red and blue. The reason is because red is only 4 away from this carbonyl. Blue is only 4 away from this one. I can cross those out. I'm just going to put a cross. Those aren't going to work. Now it's between black and green. Black is 6 carbons away, so this looks like it's a possibility. Green is 1, 2, 3, 4, 5, 6 carbons away as well. But we have an issue, which is that 3 of the carbons within this chain are also in a ring. If I use the green one, I'm going to wind up getting a bicyclic product. Ideally, I'm trying to avoid using multiple atoms in my chain that are also in a ring. Notice that if I use look at the difference. If I use black, how many of my atoms are in the ring? Just 2. I have one; let me start. I have too many numbers going on now. I hope you guys are learning about this. If I start from black, I have 1, 2, 3, 4, 5, 6. Notice that only 2 of the atoms are within the ring. Now if I start from green, 1, 2, 3. The first three atoms of that chain are in the ring. I'm going to wind up getting a bridge compound. It's going to be kind of difficult to draw. Let's draw the other one instead. The other one is going to be more stable. For this compound, because I have a ring involved, I would recommend against doing my system where you draw a cyclohexane and then number it. I would try to rearrange this so it looks more like the enone that it's going to make soon. I'm going to rotate this bond. If you rotate this bond, it's going to look more like what it will eventually look like. I'm going to draw it like this now. I've got this molecule here,Doul bond O. But now I've got my methyl here and my carbonyl here. Notice that all I did was I rotated it. This is much better because if my negative is here and my positive is here, I see that I can just do this. That's going to be my 6-membered ring. What I'm going to get is a 6-membered ring that looks like this, a new 6-membered ring that has another 6-membered ring on it. This is the fun part about using Michael reactions. What are we missing on this thing? This ketone still exists. Nothing happened to that. Here, I should actually have an O− but that's going to get protonated to an alcohol. Eventually, this is going to be dehydrated. The direction it gets dehydrated is towards the alpha-beta. That means that my enone is going to form right here. That's going to be the final product of this reaction, my cyclic enone. As you can see, this is one of the hardest reactions in organic chemistry. It's difficult to visualize. It's kind of bulky, clunky, difficult to even conceptualize sometimes. I hope that I've made it a little bit more clear. I'm really trying to do this in a stepwise fashion, so that every step of the way, you're like, I guess that makes sense. You're not completely lost. But at the end of the day, Robinson emulation is a product that you'll have to practice drawing because you have to get good at it. All right, guys, so that's it for this topic. Let's move on.
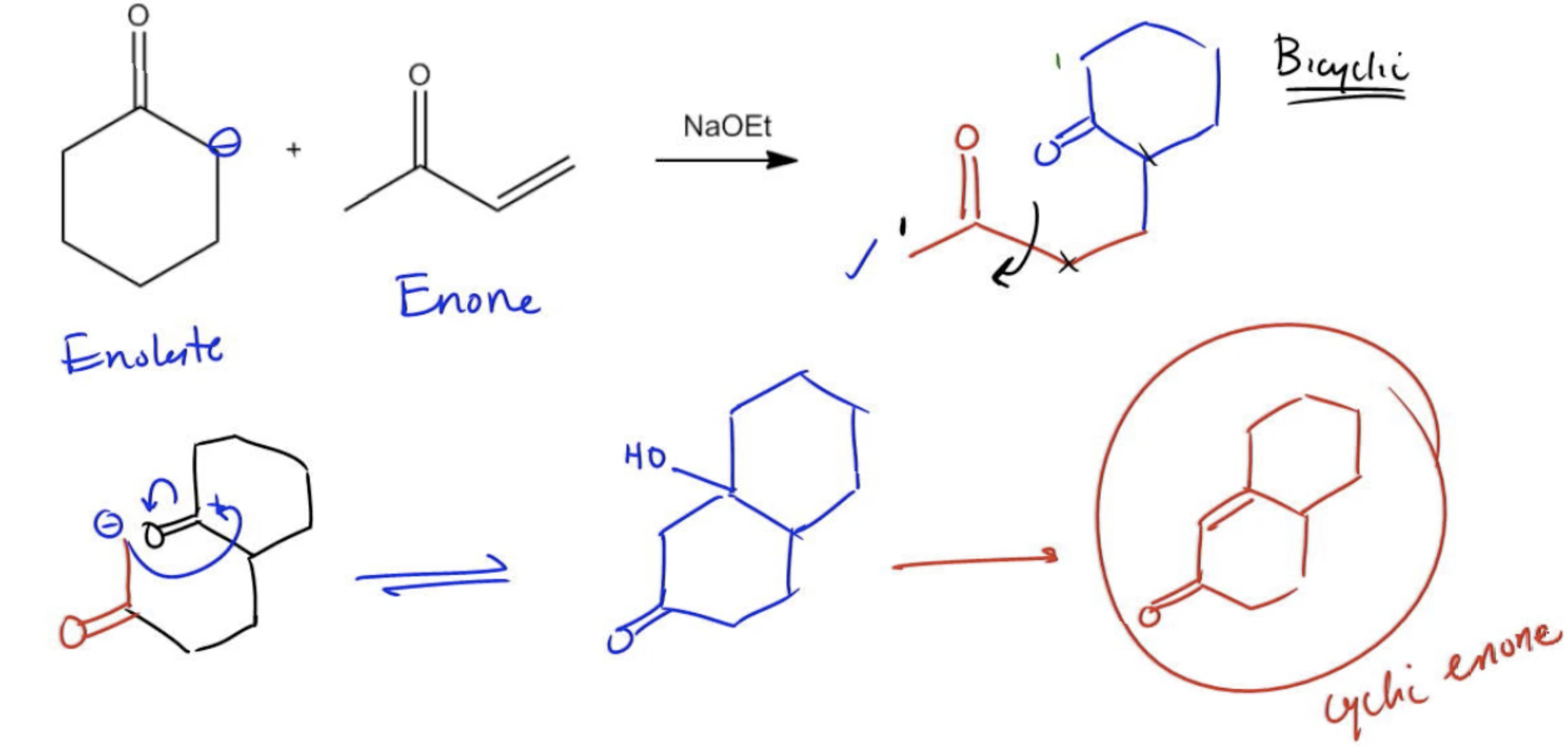
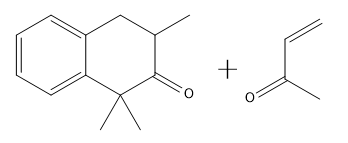
Provide the product for the following Robinson Annulation Reaction.
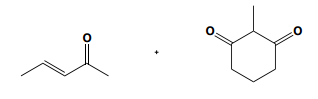
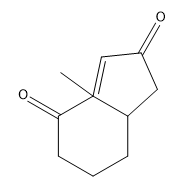
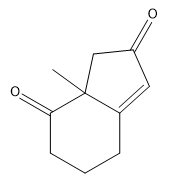
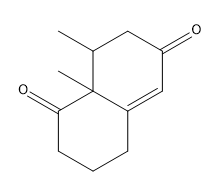

If you noticed a methyl group that vanished around 3:50 in the above video, you are not alone! Place a methyl there in your final answer and you will be good:)
How would you prepare the following compound using a Robinson annulation reaction between a di-ketone and an alpha, beta unsaturated ketone?
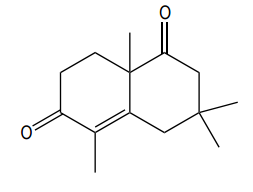

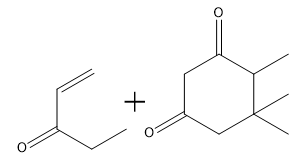
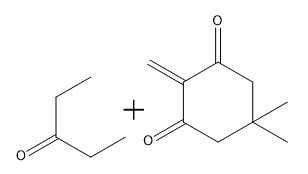
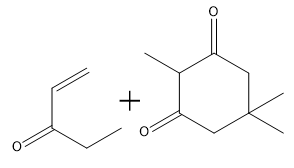
How would you prepare the following compound using a Robinson annulation reaction between a ketone and an alpha, beta unsaturated ketone?
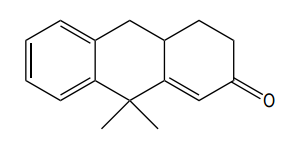

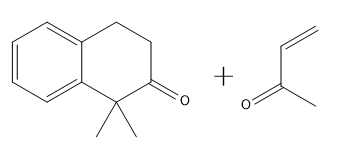

Great job, guys! You survived.
Do you want more practice?
More setsYour Organic Chemistry tutors
- Draw the product obtained by heating each pair of ketones in a basic solution. a.
- Draw the product obtained by heating each pair of ketones in a basic solution. b.
- What two carbonyl compounds are needed to synthesize each of the following compounds, using a Robinson annulat...
- Show how you would use the Robinson annulation to synthesize the following compounds. (a)
- Propose a mechanism for the following reaction. < of reaction>
- Propose mechanisms for the following reactions. (c) < of reaction>
- Show how you would use the Robinson annulation to synthesize the following compounds. (b)
- Show how you would use the Robinson annulation to synthesize the following compounds. (c)
- Predict the product of the following reactions.a. <IMAGE>
- Predict the product of the following reactions.b. <IMAGE>
- Show how the following molecule might be synthesized using the Robinson annulation.<IMAGE>
- Propose mechanisms for the following reactions.(d) <IMAGE of reaction>
- The base-catalyzed reaction of an aldehyde (having no α hydrogens) with an anhydride is called the Perkin cond...
- The base-catalyzed reaction of an aldehyde (having no α hydrogens) with an anhydride is called the Perkin cond...
- Show how you would use Robinson annulations to synthesize the following compounds. Work backward,remembering t...
- Show how you would use Robinson annulations to synthesize the following compounds. Work backward,remembering t...

