Hey everyone. In this next page, we're going to be focusing on a special type of condensation called an intramolecular condensation. All right, guys. It gets a little bit more complicated. Dicarbonyl compounds have the ability to self-condense through cyclization if it will generate a 5 or 6 membered ring. Here's the deal. Any ring below 5 members, 4, 3, way too unstable to cyclize on its own. You're going to need to put a lot of energy into that system to make it happen. In order for these reactions to be favorable, we're going to try to make 5 and 6 membered rings. That's actually going to be a theme later on when we talk about other reactions that condensation products can undergo. Here, I've got this diketone. This could happen to diesters, diketones, whatever, dialdehydes. The biggest point here is that when you make your enolate, you want to make your enolate in a place where if you attack the carbonyl, you'll make either a 5 or a 6 membered ring. Let me ask you a question. I've actually got 4 different positions that could make enolates here. I've got position 1, position 2, position 3, and position 4. You guys understand that positions 3 and 4 are the same thing as 1 and 2, so I'm going to erase them because that's just distracting me. Literally, I just have a choice between do I put the enolate on carbon 1 or on carbon 2. They both have alpha hydrogens, so I could definitely get enolates there. The question is the real way that you decide this is you decide by the ring that you could make after you cyclize. Let's look, for example, at enolate 2. If I were to put my enolate on carbon 2, then it would attack this carbonyl here. How big would that ensuing ring be? This being my first, my nucleophile always gets the 1st carbon in the count, so this would be 1, 2, 3, 4. You always count from nucleophile to electrophile. Is that big enough? No way. Let's talk about 1. If I were to make it on 1, it can't attack that one. That's way too close. It would attack this carbonyl. Then let's count. Let's see if it's the right size. 1, 2, 3, 4, 5. That is a lot better. That's a 6 carbon chain that is going to connect between the 1 and the 6, making a 6 membered ring. The NaOAT is going to specifically make an enolate here so that it can cyclize on its own. The mechanism is just going to be what you expect for any aldol reaction. You would kick up the electrons to the O and then it would protonate. What we could do here is what I like to do, if we already know it's going to be a 6 membered ring, let's just draw a 6 membered ring and then let's just add the groups. From lots of years of experience, I know that it's actually going to be easiest to number this ring if I start at the top left corner and I work my way around counterclockwise. 1, 2, 3, 4, 5, 6. Literally, guys, this is just me trying to make it look as pretty as possible. You could have numbered it however you want. As long as your numbers are in order, it doesn't matter. The only thing you can't do is like 1, 3, 5, 7, or whatever. What am I missing? What groups do I need? It looks like I'm missing a ketone on 2. I'm going to put that there. What else am I missing? It looks like I'm missing some stuff on 6. What am I missing on 6? Could you guys tell me exactly? Think about it. Think about exactly what's not on this chain, on this ring. I'm missing an OH because we said that it's going to protonate. Am I missing anything else? I'm also missing a methyl because this methyl was not part of the ring. It's just extra. There we go. Now we have it drawn correctly. This is called what? What's the name of this product? This is our beta-hydroxycarbonyl. You're getting professional at this at this point. But what do we know about beta-hydroxycarbonyls? They love to dehydrate. We're going to dehydrate this bad boy and we're going to dehydrate between the alpha and the beta. What this is going to make as a final answer is I'm going to get what we call a cyclic enone. This is going to be highly favored because that's really stable. You're going to get a cyclic enone and that's what you get when you have basically an intramolecular reaction that's an aldol. This would be specifically an intramolecular aldol. You might say, Johnny, how do you know it was aldol? Because it's ketones or aldehydes. You're going to get a cyclic enol. In the next video, I'm going to show you what happens for intramolecular condensations of esters.
Intramolecular Aldol Condensation - Online Tutor, Practice Problems & Exam Prep
Dicarbonyl compounds have the ability to self-condensate through cyclization. When a diketone or dialdehyde self-condensate, the resulting product is called a cyclic enone.
Diketones
Video transcript

When a diester self-condensates, the resulting product is called a cyclic β-ketoester.
- This is known as a Dieckmann Condensation or Intramolecular Claisen
Diesters (Dieckmann Condensation)
Video transcript
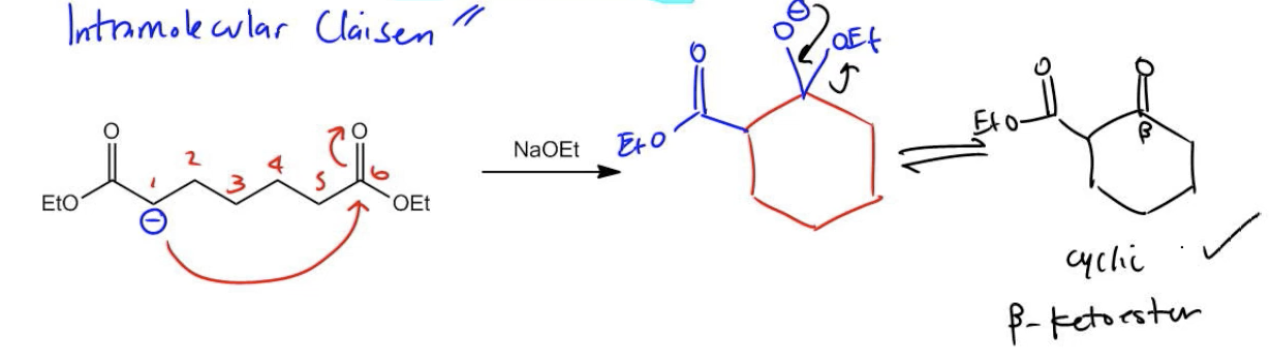
When a diester cyclizes and self-condenses, the product is going to be a cyclic beta-ketoester. This just makes sense because a beta-ketoester is always the product of a Claisen. But now it's going to be a ring because it's intramolecular. Chemists just had to make this overly complicated and, apparently, the person who discovered this specific cyclization was called Deichmann. This is also called a condensation. But don't get too hung up on the name because really all it is is an intramolecular Claisen. Intramolecular Claisen and Deichmann are the same thing. Once again, we're going to determine where to form enolates based on whether we should, based on if we can make a 5- or 6-membered ring. Esters are a little bit more straightforward because I don't have possibilities on both sides. I only have really 1. Would that enolate make sense? Would that give me a 5- or 6-membered ring if it attacked the other ester like this? Let's see. That's perfect. A 6-membered ring, we know that that's going to be favored.
What I'm going to do is draw the rest of the mechanism which would be O facing up. Once again, since I have a 6-membered ring, I'm going to draw my six numbers. I'm going to start at this corner and I'm going to move my way around counterclockwise. Really, you can start wherever you want but this is just the way that I like to do it. Now I'm wondering what's missing in the different places. Let's even just do the whole mechanism. At this point of the mechanism, what's missing? It looks like 1 is missing an ester. Let's add that. 1 is missing this whole ester thing. What else? 6 is missing two things. 6 is missing an O-negative and it's missing an O18. Perfect. Now that looks like it's correctly drawn. I'm going to erase the numbers because we know we did it right.
Now, the next step of this mechanism is? What's the next step? The next step is that I have to kick out the leaving group. I would reform the ketone and kick out the OET. What that's going to give me is a compound that now looks like OET and then ketone. Now, I'm wondering, guys, did I get the right compound? Because remember that whenever you get a Dieckmann, a Dieckmann should always yield a cyclic beta-ketoester. Is this a cyclic compound? Yes. Is it a beta-ketoester? Well, I still have an ester and I have a ketone at the beta. So yes, this is correct. Cool, right? So intramolecular. Got to get used to it in this chapter. This isn't the last time it's going to come up. It's going to come up again. I'm warning you. Get good at it now. Let's move on to the next topic.
Consider the following reaction. Provide a stepwise mechanism to explain the given transformation.

Problem Transcript
Do you want more practice?
More setsYour Organic Chemistry tutors
- Draw the product of the reaction of each of the following compounds with a base: c.
- Can 2,4-pentanedione undergo an intramolecular aldol addition? If so, why? If not, why not?
- Draw the product of the reaction of each of the following compounds with a base: d.
- The following compound results from base-catalyzed aldol cyclization of a 2-substituted cyclohexanone. (b) P...
- The following compound results from base-catalyzed aldol cyclization of a 2-substituted cyclohexanone. (a) S...
- When cyclodecane-1,6-dione is treated with sodium carbonate, the product gives a UV spectrum similar to that ...
- Show how octane-2,7-dione might cyclize to a cycloheptenone. Explain why ring closure to the cycloheptenone is...
- Draw the product of the reaction of each of the following compounds with a base:a. <IMAGE>
- Draw the product of the reaction of each of the following compounds with a base:b. <IMAGE>
- Propose mechanisms for the two Dieckmann condensations just shown.<IMAGE of reactions>
- Predict the products of the following aldol condensations. Show the products both before and after dehydration...
- Predict the products of the following aldol condensations. Show the products both before and after dehydration...
- Show how each compound can be dissected into reagents joined by an aldol condensation, then decide whether the...
- Propose mechanisms for the two Dieckmann condensations just shown.<IMAGE of reactions>

