So like I said, like Newman projections, there's actually a lot of different types of projections. As you can see, this one right here is called a Fischer Projection. It's used mostly for sugars. So later on, if we get into sugar chemistry and carbohydrates, we'll be using Fischer Projections a lot. But they're also used in this chapter as well. Two other common ways that are used are the Haworth projection. The Haworth projection is just that 3D projection of a ring. That's the actual name, and it's usually used for rings. So it's used to highlight what's at the top and what's at the bottom. And then finally, we have this one called a Sawhorse Projection, and this one is usually used for stereochemistry to basically say how these atoms are related to each other in terms of their orientation, their shape, their configuration. Well, in all of these cases, whichever projection we're using, also remember there's Newman as well, in all of these cases, we're going to have to convert them into bond line before analyzing them completely. What that means is that these projections are really good for analyzing certain types of things. But if we want to compare them against other normal molecules, we're going to have to convert them into bond line first because that's really like our metric system. That's our standardization.
Fischer Projection - Online Tutor, Practice Problems & Exam Prep
There are several common projections used to visualize molecules in different perspectives.
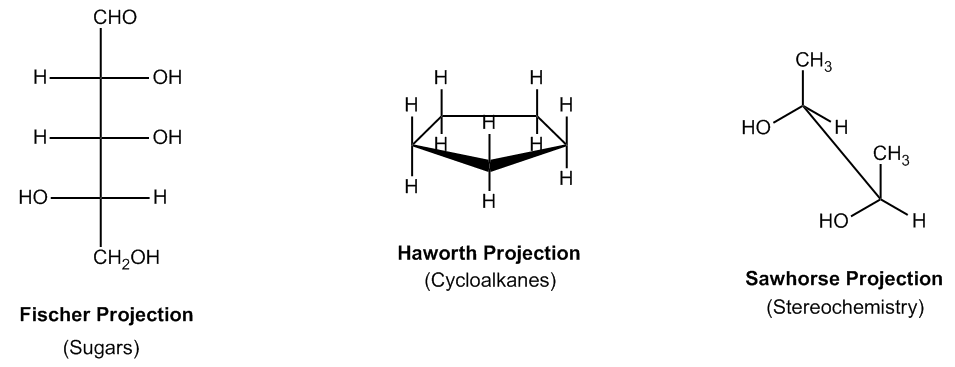
Introduction to different projections.
Video transcript
We will need to know how to convert these into bondline so that we can analyze them.
How to convert Fischer projections into bondline structures
Video transcript
So basically, you can see I have made a caterpillar. What am I talking about? Well, really, a Fischer projection, even though it looks 2D, it's not really 2D. The way it's really supposed to be interpreted is that every vertical bond is going into the page on a wedge. Okay? So these would be vertical and vertical. And then every horizontal bond coming off of it is a wedge. I'm sorry, I might have said wedge. Those are dashes and these are supposed to be wedges. So even though it looks 2D, it's really not 2D. That's just the way it's drawn, to make it easier. But really the way it's supposed to be interpreted is with wedges and dashes. What I asked you guys to do is, okay, if we're going to convert this into a bond line, we need to use this wedge and dash notation first. So, if you're given a bond line, first do what I just did and make it wedge and dash, like I just did. Then we're going to use an eyeball and we're going to pretend like we're looking at it from the side. And what we're going to see is that it's going to make what I call a caterpillar. Basically, what that means is that this CHO here would be right here, CHO. This CH2OH would be here, CH2OH, and what we would notice is that we have 3 different junctions, 1, 2, 3, and these are the places where bonds come off the top. And if you think about it, maybe draw a line down here, this actually kind of looks like a caterpillar. Right? Now this just got really goofy, but it kind of looks like a caterpillar with it's like it's like on a leaf and it's like eating away and it's like munching away, and it's got like its little hair sticking up. That's why I call it a caterpillar. Alright? So basically, I don't want you guys to necessarily draw the whole caterpillar. You don't need to draw a face or anything. I'm not going to be strict about that. But what you should do is realize that this bond here, 1, relates to one right here. So what that means is then I look at the eyeball and I say okay, according to that eyeball, what should be in the front and what should be in the back? What does it seem closest to itself? And what it's seeing is that there should be an H in the front, because that's the closest one to the eyeball, and there should be an OH in the back, because that's the furthest one from the eyeball on the one carbon. Is that making sense? So I'm looking at the one carbon and saying what's in the front, what's in the back. In the same way I would work with the other ones. Then I'd say 2 should have an H in the front and an OH in the back, and then 3 should have an OH in the front and an H in the back. Once I have my caterpillar, then I have to do my last step and that's going to give me my bond line. So we're actually really close to the bond line. The thing is that bond line structures, are they ever like that where all the bonds are in a straight line? Usually not. Usually there's a zigzag pattern. Right? So we need to restore this back to a zigzag pattern. How do we do that? By rotating every other bond. And another thing I like to say is that by rotating every even bond. So what we're going to do here is I'm going to show you guys how to do this. Basically, what we would do is we have 1, 2, and what we would do is we would rotate every other bond. So we would rotate 2 as my atom. I'm going to want 2 to face down. And if we rotate 2 to face down, that's going to restore my zigzag. So now what I'm going to do is I'm going to draw this like this, where I have 1 is here, 2 is here, 3 is here, and then it goes down like this. So then the CHO is in the same exact place, The CH2OH is in the same exact place. In fact, 1 and 3 are in the same exact place. Notice that they were both pointing up before. Before, 1 and 3 are both pointing up. So that means that the groups that are on 1 and 3 should look exactly the same. That means that 1 should have an OH at the back and it means that 3 should have an OH at the front. Do I have to draw the H's? No, because this is bond line. Remember in bond line, H's are omitted. The only thing that's changed is that now I'm rotating 2 down. That means that whatever I had on 2 has to flip. Where should the OH go? Should it go on the back, the front? Since it's rotating, the OH should now go on the front. Because of the fact that it rotated down, that means that that bond that was in the back is now going to rotate to the front. So now what I've just done is I've just made my bond line. That is a bond line structure right there. And all I did was I rotated every other bond, so meaning that I rotated this one. This one doesn't get rotated. See, like this one is fine. That one doesn't get rotated, But then this one got rotated and then this one didn't. So notice that every other one I rotate and if this is a longer chain, then I would have also rotated atom 4 to go down. So I would have rotated atom 2, atom 4, atom 6, until my Fischer projection is done. And that would make the zigzag pattern. What I want you guys to do is just as a free response, convert the following Fischer projection into a bond line structure, go ahead and try to solve it yourself, and then I'll go ahead and step in and show you guys how.
- Make a caterpillar, then rotate every other bond.
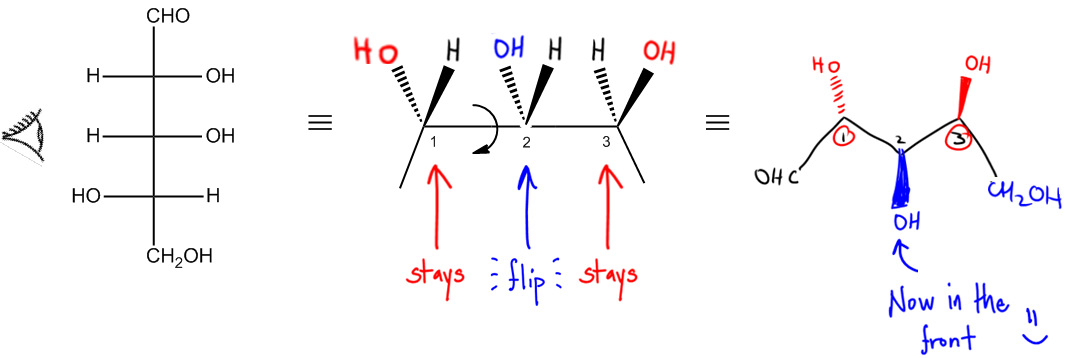
Convert the following Fischer projection into bondline structure.
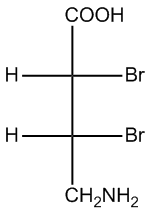
Convert the following Fischer projection into bondline structure.
Video transcript
Alright, guys. Let's go ahead and go step by step. The first step would be to redraw this with the wedge and the dash. So I would put this on a dash. I'll put this on a dash. I would put these guys on wedges. Br, Br, H, H. Is that cool so far? Now what I want to do is I'm going to draw my eyeball because that's going to help me remember what things look like. Okay? Notice that I have carbon 1 here, carbon 2 here, these are the back of the caterpillar. So now when I convert this into a caterpillar, what it's going to look like is like this, where I have atom 1, atom 2, that's the back of the caterpillar. What it's going to have here is COOH. What it's going to have on over here is CH2NH2. Now I just have to figure out what's on the top, what's on the hairs. Okay? So it should actually be really simple. I should just have for my wedges, I should have H and H. Okay? Because those are the ones that are closest to the eyeball. For the back, what I should have is BrBr. Is that making sense so far? Cool. Now, I think a question that some of you guys might have is that notice that before up here, I was drawing the H's on the right side and the front, the wedge on the right and the dash on the left. And then here I was drawing the wedge on the left and the dash on the right. It does not matter. You can draw them however you want, as long as the thing that's in the front is still in the front, the things that's in the back is still in the back. Alright? So now we have our caterpillar. So now how do we convert this into a bond line? All I do is I rotate every other bond or what I want to make sure is every other atom is face down. Every even atom is face down. So that means that's going to be atom 2. Okay? So atom 2 is going to be the one that has to face down. Okay? So let's go ahead and convert this. What that means is that now this is going to turn into this, this and that, where this is now atom 1.
Do you want more practice?
More setsYour Organic Chemistry tutors
- Convert the Fischer projection to a perspective formula.<IMAGE>
- Convert the Fischer projection to a perspective formula.<IMAGE>
- Convert the following perspective formulas to Fischer projections. a. b.
- For each Fischer projection, make a model. 1. draw the mirror . 2. determine whether the mirror is the same ...
- For each set of examples, make a model of the first structure, and indicate the relationship of each of the ot...
- Convert the following Fischer projections to perspective formulas a. b.
- Convert the line-angle drawings into Fischer projections. (c)
- Convert the line-angle drawings into Fischer projections.(a) <IMAGE>
- Draw Fischer projections of the following molecules. (b)
- In this attempt to convert the line angle drawing of d-erythrose (shown) to the Fischer projection (shown), by...
- Draw Fischer projections of the following molecules.(c) <IMAGE>
- Convert the following perspective formulas to Fischer projections.c. <IMAGE>d. <IMAGE>
- (a) There is only one ketotriose, called dihydroxyacetone. Draw its structure.(b) There is only one aldotriose...
- For each set of examples, make a model of the first structure, and indicate the relationship of each of the ot...
- Which of the following compounds are chiral?Draw each compound in its most symmetric conformation, star (*) an...
- Draw a Fischer projection for each compound. Remember that the cross represents an asymmetric carbon atom, and...
- a. Are d-erythrose and l-erythrose enantiomers or diastereomers? b. Are l-erythrose and l-threose enantiomers...
- For each pair, give the relationship between the two compounds. Making models will be helpful. e. and f. ...
- For each structure, 1. draw all the stereoisomers. 2. label each structure as chiral or achiral. 3. give the r...
- Give the stereochemical relationships between each pair of structures. Examples are same compound, structural ...
- Are the following pairs identical, enantiomers, diastereomers, or constitutional isomers?c. <IMAGE>d. &l...
- For each pair, give the relationship between the two compounds. Making models will be helpful.g. <IMAGE>...

