Hey everyone. So now that I know in general terms how to predict what types of stereoisomers I'm going to get based on the number of chiral centers, I'm going to start teaching you some of those exceptions to the rules. And the first of those exceptions is a chiral compound that doesn't have any chiral centers at all, and these are called atropoisomers. Okay. So atropoisomers are these unusual compounds that contain no chiral centers, yet they actually are chiral due to their inability to freely rotate. Now it really depends, class by class, professor by professor, how much emphasis they put on these compounds because they are unusual and they're not ones that we deal with a whole lot in Orgo 1. But sometimes your professor might just throw these in as a trick so that you'll forget about these kinds of compounds that can't rotate. Okay? So here I have just a few. In fact, there's a few more, but these are the really common ones. So allenes. As you can see, allenes, they're two double bonds together. They can't rotate because double bonds can't rotate. Substituted biphenyls. I know you don't know what that is yet, but a biphenyl is just two phenyl groups that are attached in one spot and they're substituted, meaning that they can't really rotate because those substituents that you can see sticking out kind of act as teeth. They lock it together, so it can't fully rotate. Okay. Trans cyclooctene. This is actually a molecule that I was just recently talking about how it can form the cis version and the trans version. It turns out the trans version, because it can't rotate, it can actually form a right-handed version and a left-handed version of this twisted, messed-up thing, and is chiral because of the lack of rotation. Then finally, BINAP, which I don't even remember exactly what it stands for, but it's very similar to the substituted biphenyl where except in this case, one of an entire phenyl group or an entire benzene ring is acting as one of those teeth that are not going to let it rotate across that single bond. So, all of these are going to be chiral even though they lack chiral centers. These are, like I said, this is not the usual. This is not the norm. These are unusual molecules that happen to display chirality.
Atropisomers - Online Tutor, Practice Problems & Exam Prep
These are exceptions to the rule:molecules that contain no chiral centers yet are chiral due to their inability to freely rotate.
Recognizing chiral molecules with zero chiral centers.
Video transcript
Allenes
Determining if allenes are chiral or not.
Video transcript
So what I want to do is teach you guys tests for the two most common ones, which are allenes and substituted biphenyls, and the other ones don't really need rules, so you're fine. You can just always assume that they're going to be chiral. However, it turns out allenes and substituted biphenyls can be chiral, or they cannot be chiral depending on the rules they follow. So let's look at this. We are simply going to use a modified version of test two, which is the one for stereo centers, to identify trigonal centers in the allene. But you might wonder about the two double bonds. Well, here is the tricky part: we are going to visualize the allene as just one big double bond. Maybe squint a little bit and try to see it; try to ignore the carbon in the middle and just pretend it's one big double bond. By the way, this is just my way of solving these, but I think it has helped a lot of students, and I believe it will help you too.
If it can form E or Z isomers after visualizing it as a big double bond, that means it's chiral. Remember when I taught you guys about trigonal centers, I mentioned that they're actually achiral if they pass the test. But allenes are different, so just think about allenes as their own thing. Allenes are going to be chiral if they can form E or Z through this weird long double bond. So, I'll do the first one with you guys, and then I'll let you guys do b and c on your own.
Here's what I would do: look at this compound and ignore the "c" in the middle, just pretending it's one big double bond. Then, I would ask myself if this double bond is able to form E and Z, or cis and trans. Since I have two of the same atom on one carbon, on one side, it actually cannot form E and Z, or cis and trans. This means that no matter how much I switch these two groups, I'm never going to be able to get E and Z. So, this one would be achiral. Now, I want you guys to solve further and use this knowledge to determine the chirality in other cases.
Imagine that the allene is a big double bond. If it is able to form E or Z isomers, it is chiral.
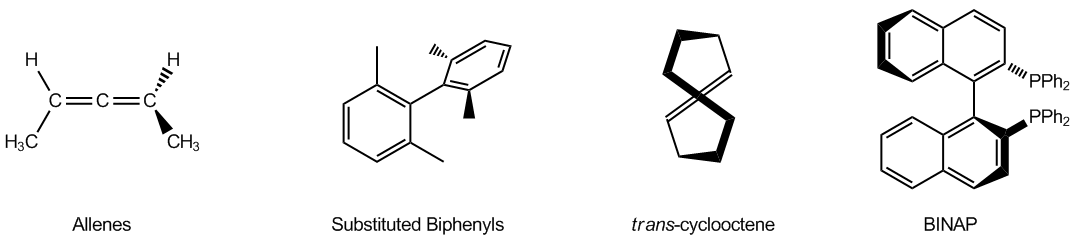
Is the following allene chiral?
Is the following allene chiral?
Video transcript
Alright, so it is going to be chiral. Okay. And the reason it's going to be chiral is because if I visualize this as a big double bond, okay, then what I have is that none of the carbons have two of the same group on them. So that means that if I were to draw my fence here, my methyl groups could arrange in two different ways. This would be that they're both on the same side, so this would be the cis conformation, but I could also have a situation where my allene looks like this, where I have my CH3 and my H, but then my CH3 is over here and my H is back here. Okay? And that would be the transversion. Now like I said, is this chemically accurate? Not really. But it's going to help you guys to figure out which ones are chiral or achiral. So this one would be chiral. Go ahead and try to do.
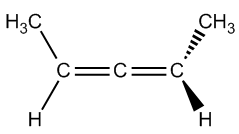
Is the following allene chiral?
Is the following allene chiral?
Video transcript
So hopefully you said that C is achiral. Okay? Because C, if you visualize it as a big double bond, has 2 of the same group on one side. So that means that no matter how much I rotate these guys here, no matter how much I flip them, I'm always going to have the same compound. So this one again, since that's 2 of the same thing, just like this situation over here, which is a bad situation, they're both going to be achiral because they can't form cis and trans. Alright, so that was easy. Right? So now what I want to do is move on to substituted biphenyls.
Substituted Biphenyls
Is the following substituted biphenyl chiral?

Determining if substituted biphenyls are chiral or not.
Video transcript
So these guys are going to have a pretty weird rule too. Basically, for substituted biphenyls, what we look at is the substituents in the ortho position. Now, ortho is a word we're not really going to use until organic chemistry too very much, but just as a heads-up, what it means is next to or adjacent to. Okay. So basically what we're looking at is the substituents that are next to that single bond or that sigma bond that it's rotating around. Okay? And what I want to do is I want to figure out if any of the rings have 2 of the same group. If they have 2 of the same group, then it's not chiral. But if it has 2 of the same group, then that's going to be achiral. Okay? But if it has 2 different groups, then it's fine, then it would be chiral, similar to the way that we did it for alanines. Okay. So let's go ahead and do together, and then I'll have you guys do B and C on your own. So for A, do I have 2 of the same groups on any of the rings in the ortho position? The answer is no. These groups are different, and these 2 groups are different. So that means this is going to be a chiral substituted biphenyl because none of the rings have 2 of the same exact thing on one side. So go ahead and try to figure out B and C.
Must meet two criteria to be chiral:
- All substituents are in the ortho- position
- None of the rings have two of the same group on them
*ortho- is a term we'll use in Organic Chem 2. It basically means that a group is adjacent to the primary carbon linking the two rings.
Is the following substituted biphenyl chiral?
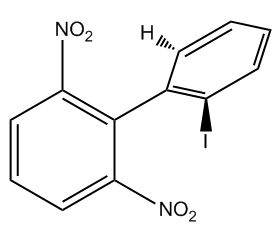
Is the following substituted biphenyl chiral?
Video transcript
B is achiral. Why? Because it has two of the same group on one of the benzene rings. Okay? Just so you guys know, this is a group that we're not going to deal with a lot in organic chemistry 1. This is called a nitro group. Okay. Just introducing it in case you guys are probably going to see it in organic chemistry 2. Okay. So, a nitro group, but there are two of the same ones, so it doesn't matter how unusual it is. If there are two of the same thing, it cannot form cis and trans. That would be achiral. Let's move on to C.
Is the following substituted biphenyl chiral?
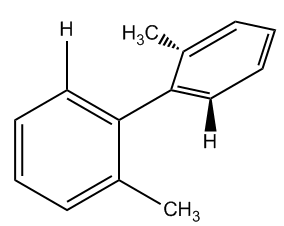
Is the following substituted biphenyl chiral?
Video transcript
So I'm going to take myself out of the screen here, so you guys can see everything. So in this case, do I have 2 of the same group on this molecule or on any of these rings and the answer is no. This one, I have 2 different groups. This one, I have 2 different groups. So all I'm looking at is I don't have 2 of the same ones, so this one would be chiral. So hopefully that made sense to you guys, and let me know if you have any questions on atrope isomers. Remember that these are like a specific subset of molecules, but are not the norm. So you should really use the general rules that I told you about. If it does not have a chiral center, assume that it's achiral unless it's one of these 4 guys that I talked about. Alright, so let's go ahead and move on to the next topic.
Do you want more practice?
More setsYour Organic Chemistry tutors
- The original definition of meso is 'an achiral compound that has chiral diastereomers.' Our working definiti...
- Draw three-dimensional representations of the following compounds.Which have asymmetric carbon atoms? Which ha...
- (••••) A compound with two chiral centers that is meso will always have opposite absolute configurations at th...
- Which of the following compounds has a stereoisomer that is a meso compound? C 2,4-dimethylpentane D 1,3-dichl...
- Which of the following compounds has a stereoisomer that is a meso compound? A 2,4-dibromohexane B 2,4-dibromo...
- Which of the following has an achiral stereoisomer? A 2,3-dichlorobutane B 2,3-dichloropentane
- Which of the following compounds are chiral? Draw each compound in its most symmetric conformation, star (*)...
- Which of the following compounds are meso?(e) <IMAGE>
- Which of the following compounds are meso?(g) <IMAGE>
- For each structure,1. star (*) any asymmetric carbon atoms.2. label each asymmetric carbon as (R) or (S).3. dr...

