Hey guys. In this video, we're going to talk about an extremely important way to get rid of carboxylic acids, and that's called decarboxylation. Decarboxylation is going to be a very important reaction for the rest of organic chemistry. It's really important that you understand how to do it now. The way that it works is that a beta carbonyl carboxylic acid can decarboxylate in the presence of heat. What this does is it produces CO2 gas as a byproduct. It evolves out of the material. What a beta carbonyl carboxylic acid is, it's just that you have some kind of carbonyl beta to your carboxylic acid. When you heat that up, what's going to wind up happening is that you literally cleave the entire carboxylic acid off and it becomes CO2 gas, leaving only the carbonyl portion behind. Obviously, we need to get to the mechanism for this because it doesn't really make sense just drawn like that. Let's do the mechanism. For this mechanism, it's going to make a lot more sense if you do one thing: if you rotate the bond. We're going to rotate this bond up when I draw it because it's going to make more sense with the mechanism. I'm actually going to draw my OH facing like this and my carbonyl down. But this is still the same exact molecule. I just rotated it. And you should too. When I draw this mechanism, it's actually a series of 4 arrows, and there's no right place to start or no right place to end. It's all concerted. But I like to start from the oxygen grabbing the H because that's kind of like an easy place to remember for me. I would start with the O grabbing this H. It's going to start off like that. If I make a bond, I have to break a bond because H can't have 2. This single bond is going to give its electrons and become a pi bond between that carbon and that O. Now that carbon has 4 bonds. Instead of breaking the bond to the carbonyl though, I'm actually just going to break the single bond to the carbon completely. Notice what this is going to do is it's going to release my C with 2 Os on it because now there's no single bond connecting it to the alkyl group. Finally, once I make that bond, I have to break this bond. There you go. Those are the 4 arrows. This is a heat-mediated reaction. I wind up getting something that looks like this: RO2 with a double bond, and I'm going to get CO2. Now it makes sense why I get the CO2 gas. And it should even make sense why the carboxylic acid's gone. What might be a little confusing for you is that I told you that you get a carbonyl from this. Right now, I have an alcohol. That doesn't seem to make sense. But this is something that we're going to talk about a lot more if you haven't talked about it yet. But there's something special about vinyl alcohols. Does anyone remember? What's special about vinyl alcohols? They undergo this completely separate special process called tautomerization. If you want more information on tautomerization, I have an entire video for that, and there's an entire section of your textbook dedicated to tautomerization and enol chemistry. This is called an enol. Enol or enolate chemistry is a huge deal. You're going to get there if you haven't yet. But the whole point being that when you tautomerize, what you do is you switch the locations of a double bond and a hydrogen. What this actually comes after tautomerization is this. This is the keto form. You wind up getting the ketone. Just take my word for it that it tautomerizes, and then you're fine. Awesome. What I want you guys to do is for the next example, go ahead and predict what the product would look like. I know it's a big complicated molecule. Predict it, and then I'll explain what's going on.
Decarboxylation Mechanism - Online Tutor, Practice Problems & Exam Prep
Lets say you want to get rid of a carboxylic acid. Is it possible? Yes, but under certain circumstances. Let's see when that is.

General Mechanism
Video transcript
Mechanism:

Predict the correct structure
Video transcript
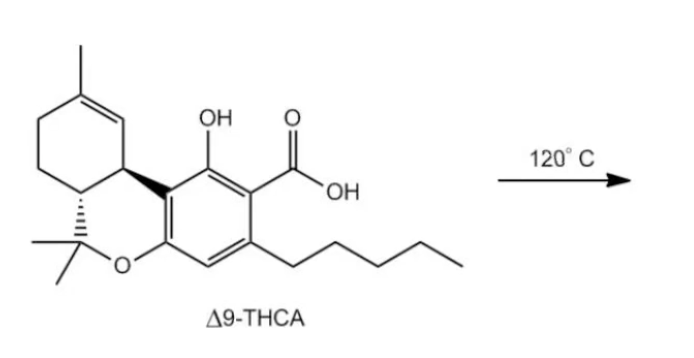
Before I say anything, we're just going to draw the product and then we will discuss. That's the annoying part, just drawing this whole thing. Just bear with me. Follow along. We're getting close. I've got this chain and that's it. Notice that when I decarboxylate, I'm going to get rid of my carboxylic acid. I'm going to need heat to do that. Does anyone know what this molecule is? This molecule is called tetrahydroquinan acid or THCA for short. This molecule is called Tetrahydrocannabidiol. I met a girl with that tattoo one time. It was awesome because it was organic chemistry. What is this? THC is the active component of marijuana. This is the good stuff, right? It turns out that what we just did was a decarboxylation reaction that actually happens in real life. Obviously not with you because it's illegal. At least in my state, it's still illegal. But where you are, we'll see what state law says. Tetrahydrocannabinolic acid decarboxylates at about 120 degrees to produce the active form of THC. THC A is an inactive form of the drug. In order for the drug to actually work, to actually do what it's supposed to do, it's going to need to decarboxylate it first which is why they actually smoke it, right? You smoke it to decarboxylate it to activate it. Anyway, I just thought that was an interesting application. It is probably the most famous application of decarboxylation in the real world but it happens all the time in all kinds of organic reactions, so good for you to know. I know you're not going to forget it now. Let's move on to the next video.
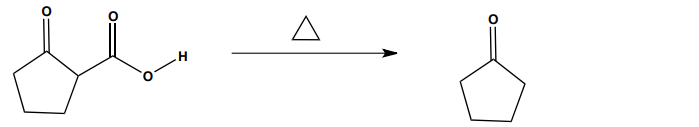
Provide the mechanism for the following reaction.
Problem Transcript
Which carbonyl group will undergo decarboxylation most readily?

Do you want more practice?
More setsYour Organic Chemistry tutors
- Which of the following compounds decarboxylates when heated?
- Which of the following compounds will decarboxylate when heated? c. d. e.
- Although metal ions increase the rate of decarboxylation of dimethyloxaloacetate, they have no effect on the r...
- 3-Amino-2-oxindole catalyzes the decarboxylation of a-keto acids. a. Propose a mechanism for the catalyzed re...
- Which of the following compounds will decarboxylate when heated?a. <IMAGE>b. <IMAGE>
- (••••) LOOKING AHEAD In Chapter 19, we will learn about the hydrolysis of t-butyl esters. In the reaction belo...
- (••••) LOOKING AHEAD While acidic conditions were used in Assessment 18.68, decarboxylation of esters can also...

