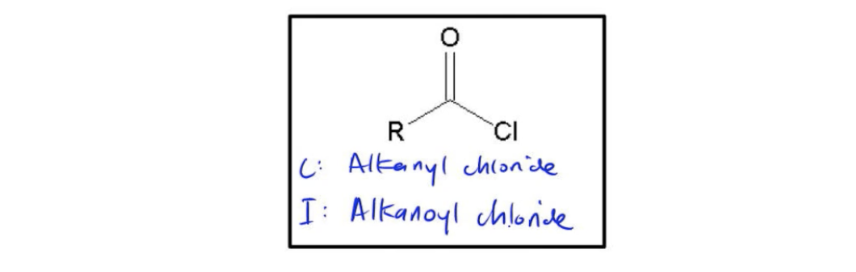
Let's name some acid chlorides. So guys, acid chlorides are not hard to name, but it's important to know that the process you use is going to change a bit depending on whether you're trying to do a common or an IUPAC name. Since there are a few rules associated with this, I think the best way to learn it is just to draw an example. So let's go ahead and say that we had a 3-carbon acid chloride. And we're trying to figure out both the common name and the IUPAC name for it. I'm actually going to move this over so I have more room to write. Perfect. So let's go ahead and start off with the common name. The rules for the common name are that you remember that the common name starts from the carboxylic acid. You're basically imagining this was a carboxylic acid. Now, how what would be the prefix you would use? What would be the root name? Sorry. So if it's 3 carbons, we know that would be propionic acid. So let's actually write that down. You're going to need your eraser because we're going to erase it, but you can just write it for now. Propionic acid. So why am I writing this? This would be the name of the molecule if it was a carboxylic acid, right? But it's not. It's an acid chloride. So how do we change it? Well, for a common name, you replace the ic ending, ic acid ending with yl chloride. So that means I would then erase acid and ic. And I would replace it with propyl chloride. See? So we're done. All you have to do is you just take your common name and you just take out the ending and replace it with yl chloride. You're done. Now for the IUPAC, the process is completely different because remember that the IUPAC route for this wouldn't be propionic acid. It would actually just be propane, right? Because it's a 3-carbon chain, 1, 2, 3. That means in the IUPAC name, we're starting from propane. So you would say I'm going to start from propane and how am I going to change it? Well, similar to IUPAC rules, you take out the E and you add a suffix. But the suffix that we're going to add is a little different. We're going to actually add an extra O to it. So instead of being yl chloride, we're going to end with oyl chloride. It actually sounds like motor oil. That's how you pronounce it. So we would erase the E, and we would say propanoyl chloride. And guys, this is just a naming convention that is very widely used. So it's something that you should be aware of. Alright, cool. So in terms of the general names for acid chlorides, remember that for common names, you're basically using your alkanoyl. It's alkyl chloride. And for your IUPAC name, it's alkenoyl chloride. Not hard at all, just a little bit tricky. Okay. Cool. So go ahead and move on to the next set of questions and see if you can name and draw the following structures.
Acid Chloride Nomenclature - Online Tutor, Practice Problems & Exam Prep
When naming acid chlorides we will use another functional group to help us get the correct answer. Are you ready to find out which one it is?
Acid Chloride Nomenclature
Video transcript
Provide the IUPAC name for the molecule
Video transcript
Everything that applied to carboxylic acids still applies to these acid chlorides. If you're naming it with common names, you have to use Greek symbols, something like that. Let's do the IUPAC first. I know you're thinking, "Oh damn, I forgot that." IUPAC, So 2 carbons. I actually gave that one to you. Man, too easy. We've got an ethanol chloride. This substituent would be a 2 hydroxy. Let's put that all together: 2-hydroxyethanol Chloride. One little minor thing that you might be thinking is, "Hey Johnny, I remember from Organic Chemistry 1 that we used to always give the alcohol priority." But, guys, those days are over. Carbonyls, in general, any carbonyl is always going to be a priority over an alcohol. Common name. The common name is going to be again our Acetyl Chloride. But it's going to be what? It's going to be alpha-hydroxy Acetyl Chloride. Very good because this is the alpha carbon. I made that one way too easy. Let's see how you feel about the next one. Go ahead and see if you can draw that molecule. By the way, if you can't recognize that symbol, that is not a 'Y.' That is a gamma. Go for it.

Draw the Acid-Chloride
Video transcript
I'm going to draw my valeryl chloride which is going to be a 5 carbon chain with a chlorine on it. Then I've got gamma ethyl. That means that I'm going to go alpha, beta, gamma and place an ethyl group in that position. That would be the way that you draw that compound. Not too bad. Let's move on to the next topic.
Do you want more practice?
More setsYour Organic Chemistry tutors
- Name the following: a.
- Draw a structure for each of the following: c. cyclohexanecarbonyl chloride k. benzoyl chloride
- Draw the structure for each of the following:h. cyclohexanecarbonyl chloride
- Provide the IUPAC name for the following molecules.(e) <IMAGE>
- Name the following carboxylic acid derivatives, giving both a common name and an IUPAC name where possible.(o)...

