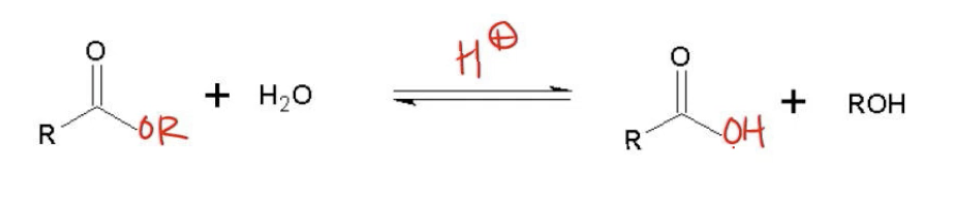
Let's talk about a reaction called acid-catalyzed ester hydrolysis. Acid-catalyzed ester hydrolysis is literally just the reverse of Fischer esterification. It's literally just the reverse reaction of producing an ester. You could then hydrolyze that ester back to a carboxylic acid. The general reaction would be that you have your ester, but you react your ester in an aqueous solution with acid and you're going to hydrolyze that ester to a carboxylic acid. Also, guys, if you recall the three rules of NAS, this would be rule number 3, the carboxylic acid conversion that says that pretty much any carboxylic acid derivative in combination with water, acid, or base could turn into a carboxylic acid. Now, what I want to do is go through the mechanism for this. By the way, I already told you it's the exact opposite of Fischer esterification. If you literally wanted to go to the Fischer esterification video and draw out every arrow backwards, every nucleophile backwards, you would get the mechanism. But I know that you guys are going to complain if I don't draw the reverse reaction. Let's go ahead and do that now. I'm going to show you guys exactly how to draw the reverse of Fischer Esterification.
Acid-Catalyzed Ester Hydrolysis - Online Tutor, Practice Problems & Exam Prep
Are you having deja vu? You should be! We are learning a mechanism that is the exact opposite of Fischer Esterification.That means instead of going from a Carboxylic Acid to an Ester, we are hydrolyzing an Ester to a Carboxylic Acid.
General Reaction
Video transcript
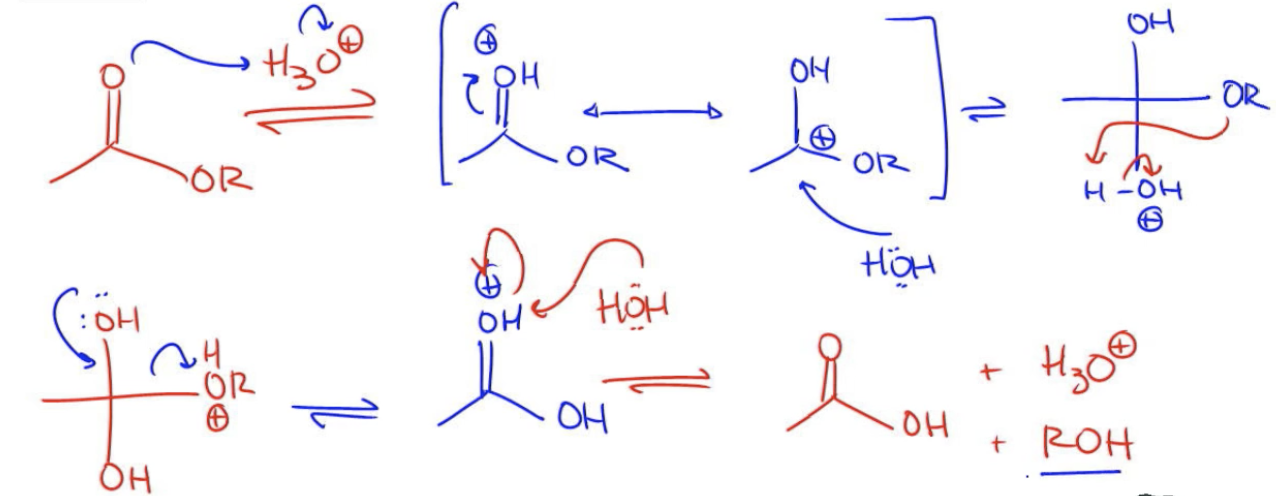
General Mechanism
Video transcript
All right. So let's start off with our ester. And I'm going to go ahead and use H3O+ as my acid. What's going to be the first step? It's going to be protonation. You got it. That's going to give me a compound that looks like this, positive charge. What's my next step? Resonate. So it's resonate. That's going to give me a positive here. My resonance structure. This is a great time to nucleophilically attack. I'm going to get water and my water is going to attack and I'm going to form a molecule. Wait, let me draw my equilibrium arrows. I'm going to form a molecule that looks like this. O H at the top, O R on the side, and water at the bottom. Can you guess what the next step is going to be? What are we trying to get rid of? We're trying to get rid of the OR which means that I want to do a proton transfer specifically to the OR this time. You might be saying, Johnny, how do you know that it doesn't go to the O? Because then that would be the forward mechanism. I'm just trying to go backwards here. I'm trying to get rid of the OR. This OR grabs that H and turns into OR+. Now what happens is I eliminate. I use the electrons from my O to kick out the OR. Oops, that's not going to work. I now have a structure that looks like this. OR+. And now I use water to deprotonate? Water to regenerate my acid. If anything, this serves as extra practice for you for the Fischer mechanism because it is that mechanism, plus my H3O+ and plus my alcohol because I generated one-equivalent of alcohol that I lost. That's really it. Let's move on to the next video.
Mechanism:
Do you want more practice?
More setsYour Organic Chemistry tutors
- Show the mechanism for the acid-catalyzed formation of 23c starting with the product obtained from its hydroly...
- Aspartame, the sweetener used in the commercial products NutraSweet and Equal, is 200 times sweeter than sucro...
- What products are formed from the acid-catalyzed hydrolysis of the following esters? a. b.
- Using the mechanism for the acid-catalyzed hydrolysis of an ester as your guide, write the mechanism—showing a...
- In the mechanism for the acid-catalyzed hydrolysis of an ester, C. what species is HB+ most likely to be in t...
- When a carboxylic acid is dissolved in isotopically labeled water (H2O18) and an acid catalyst is added, the l...
- Show how you would accomplish the following syntheses. (c) hexan-1-ol → 2-hydroxyheptanoic acid
- Suppose we have some optically pure (R)-2-butyl acetate that has been 'labeled' with the heavy O-18 isotope at...
- Propose a mechanism for the acid-catalyzed hydrolysis of phenylalanine ethyl ester.
- The Reformatsky reaction is an addition reaction in which an organozinc reagent is used instead of a Grignard ...
- Propose mechanisms for the following reactions.(c) <IMAGE>
- Propose mechanisms for the following reactions.(d) <IMAGE>
- The mechanism for acidic hydrolysis of a nitrile resembles the basic hydrolysis, except that the nitrile is fi...
- Compare each of the mechanisms listed here with the mechanism for each of the two parts of the acid-catalyzed ...
- What products would you expect to obtain from the following reactions? c. urea+water d. beta-ethylglutaric a...
- Which ester hydrolyzes more rapidly? a. methyl acetate or phenyl acetate?
- Which ester hydrolyzes more rapidly? b. phenyl acetate or benzyl acetate?
- Compare each of the mechanisms listed here with the mechanism for each of the two parts of the acid-catalyzed ...
- For each heterocyclic compound, (ii) show what compounds would result from complete hydrolysis. (a) (b) (c)
- How could you convert N-methylbenzamide to the following compounds? b. benzoic acid
- How could you convert N-methylbenzamide to the following compounds? d. benzyl alcohol
- Which alkyl halides form the carboxylic acids listed here after reaction with sodium cyanide followed by heati...
- Because bromocyclohexane is a secondary alkyl halide, both cyclohexanol and cyclohexene are formed when the al...
- What reagents would you use to convert methyl propanoate to the following compounds? c. N-ethylpropanamide d...
- Describe how the target molecule (butanone) can be synthesized in a high yield from butane. butane ?—> bu...
- Show how the following compounds could be prepared from the given starting materials. You can use any necess...
- Show how the following compounds could be prepared from the given starting materials. You can use any necess...
- Show how the following compounds could be prepared from the given starting materials. You can use any necess...
- How would you make the following compounds from N-benzylbenzamide? b. benzoic acid
- What are the products of the following reactions? d. + H2O —>
- Propose mechanisms for the following reactions. (a) (b)
- Why do the nitro groups change the relative leaving tendencies of the carboxy and 2,4-dinitrophenoxy groups in...
- Write a mechanism for each of the following reactions: a. the uncatalyzed hydrolysis of methyl propionate.
- (c) The Principle of Microscopic Reversibility states that a forward reaction and a reverse reaction taking pl...
- <IMAGE> (Solved Problem 20-1)(b) Finish the solution for Solved Problem 20-1 by providing a mechanism fo...

