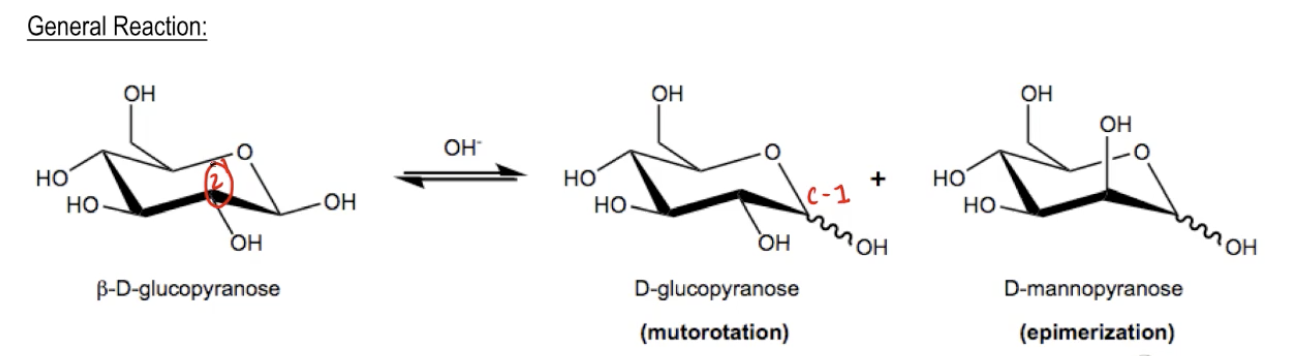
Hey guys. In this page, I want to talk about a base catalyzed reaction of monosaccharides which is called epimerization. So guys, in basic conditions, it turns out that monosaccharides can undergo a multitude of tautomerizations and isomerizations that make life really complicated. In fact, it's for this reason that we typically don't expose monosaccharides to base because it leads to so many byproducts. Let me show you one of them. So the most straightforward of these is epimerization, okay? Epimerization, remember what an epimer is, it's when you are switching just the position of one chiral center. Now when you are switching the chiral center of C1, that makes anomers and that's called mutual rotation. But when you're switching the position of C2, that's called epimerization. So monosaccharides will readily epimerize the C2 position. Several possible mechanisms exist, this can either proceed through an enolate mechanism or an enediol mechanism. I'm going to show you both of them. But let me just show you kind of the general reaction to start off with. So once again I'm just kind of picking on our beta D-Glucopyranose because it's a molecule that you should be really familiar with by now, and I'm showing you what happens in base. Well, we've already discussed mutual rotation, right? Mutual rotation is essentially epimerization of C1, okay? It's where you can get carbon 1 to form 2 anomers, to form some of the alpha and some of the beta. Okay? We've already discussed this. But it turns out that another reaction that can take place is epimerization, where now beta D-Glucopyranose can equilibrate into D-Mannopyranose. Why is that? Because it turns out that now the position can actually switch from going down to going up. How does that happen? I'm going to show you the mechanisms, but I just want to show you that both of these are going to happen at the same time and it's uncontrollable. You're going to start to get mixtures of isomers because you're going to get different anomers and now you're going to get different anomers of a completely different monosaccharide. Mannose is a different monosaccharide than glucose. That's because now the OH is going to face towards the left instead of towards the right. Also notice that in the epimerization, the anomers are also in equilibrium with each other. So it's just a big mess. Alright? So in the next video I'm going to show you the enolate mechanism and then I'm going to show you the enediol mechanism.
Epimerization - Online Tutor, Practice Problems & Exam Prep
In basic conditions, monosaccharides will undergo a multitude of tautomerizations and isomerizations. The most straightforward of these is epimerization.
General Reaction
Video transcript
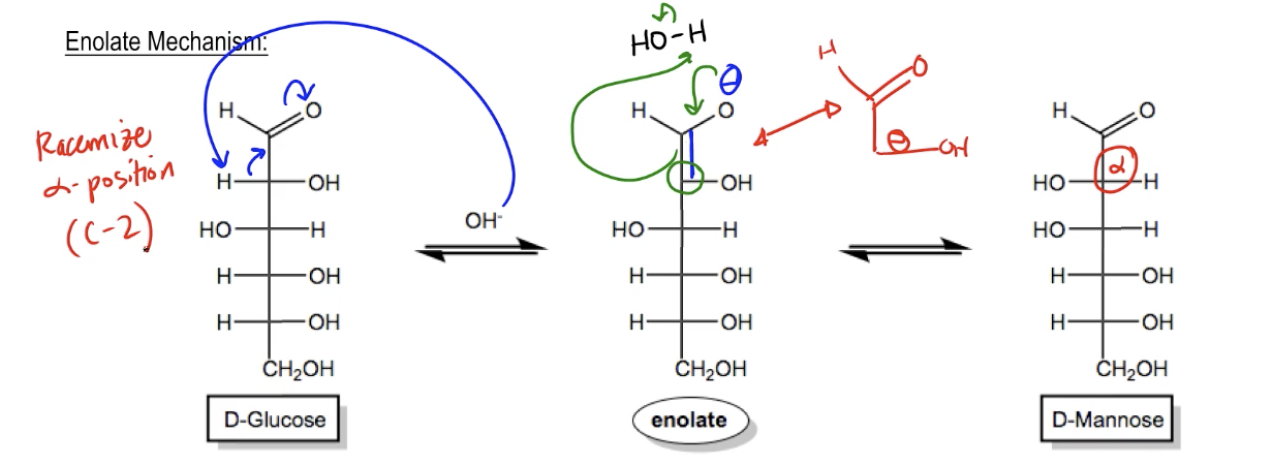
Enolate Mechanism
Video transcript
So let's go through the enolate mechanism first. And actually, my first question for you is, can you think of any reasons why specifically the C2 position of a monosaccharide is susceptible to epimerization and base? What we're talking about is this position right here. Can you think of any other principles in organic chemistry that would make that position specifically susceptible to base? And guys, we do know of a principle that would apply here and that would be the principle of alpha carbons. Remember that alpha carbons, more than usual, are uniquely acidic due to their proximity to a carbonyl. Remember that the alpha carbon is always going to have hydrogens that are much more easy to deprotonate than other hydrogens. While these other hydrogens have pKas of I don't know, you know, around 50, this guy here is going to have a pKa of around 20. Specifically, when I say this guy, I'm talking about this hydrogen right here because that's the only Alpha Hydrogen. So in the presence of base, it's not going to be difficult for my, for my O to remove that hydrogen and form and in, takes away an H, I make a double bond, and I kick electrons up to the O. So what that's going to do is it's going to form an enolate that now looks like this, double bond here, oops, no, double bond here and negative charge here. Notice that now I've lost the stereochemistry of this O. This O is now trigonal on a trigonal planar carbon, it could either be on the right or the left because it's now double-bonded. Now guys, there may be some of you that are saying Johnny that's not how I usually draw enolates, usually how I draw an enolate would just be with a double bond H and with a negative here. Is that also possible? Totally. That's the same thing. These are actually resonance structures of each other. So it's totally fine if you draw it that way. The only reason I drew it this way is because that's the way your book tends to draw it and that would be like the major contributor of the enolate. So your book tends to draw it like that but if you in principle this is the same exact reaction if you just draw the negative on this carbon because once again what's going to happen is that O is going to be able to flip back and forth because now it doesn't have stereochemistry.
Okay? Does that make sense? Awesome. So now guys, what happens? Well, now we would just protonate again. Or not protonate again, we would now protonate. So then in the next step, you have water, okay, and what you're going to do is just reform the double bond. So this negative charge is going to come down, make a double bond, and then this double bond is going to grab the H. And now it's going to become the aldehyde once again. But notice that because we lost the stereochemistry the stereochemical information on that C2 carbon, now it's possible for the O to basically racemize and go to the other side. And by the way, guys, this is not unique to sugars. One thing that we've learned in enolate chemistry is that enolates always racemize the alpha position. So essentially, nothing new is happening here. All we're doing is we are racemizing the alpha position of the sugar which happens to be the C2 position. So this totally falls in line with all the principles that we learned about enolate from enolate chemistry from your alpha carbon chapter.
Awesome guys. So that was the enolate mechanism, and next, I want to show you guys the indiol mechanism which is a competing mechanism that accomplishes the same thing.
Enediol Mechanism
Video transcript
Okay guys, so the enediol mechanism is super similar. We're going to go ahead and take away the H, make the double bond, kick electrons up to the O, this is going to give me a negative charge here and a double bond here. The only difference, guys, is that I'm now going to protonate. So I'm going to protonate that negative charge to give me literally what's called an enediol. Enediol, meaning that I have a double bond with 2 alcohols on it, enediol. So that's my enediol intermediate. Now the enediol intermediate, once again the information of C2 has been lost in terms of the stereochemical information. It could go in either direction. So I'm just going to go back to the original enolate and back to the monosaccharide. So now in this next step, I'm going to take base again, I'm going to deprotonate, and that's going to give me a negative charge here with a double bond. And then finally, I'm going to reform the carbonyl and grab an H and that's going to give me my epimer where now the OH is faced towards the other side and I now have a new monosaccharide. And basically what just happened was just another route towards C2 epimerization. You might even say Johnny like it seems unnecessary, why did you add the it's basically you're just adding a proton to take it away again and that's exactly it. But this is one of the important mechanisms, one of the other important ways that epimerization can take place. So, just you know, basically an enediol is just a longer version of enolate because in enolate I just kept it as a negative charge and went back. Here I protonated, deprotonated again, and then finally went to the epimer. Okay? Hope that makes sense. The same thing happened at the end. Let's keep moving on.

Do you want more practice?
More setsYour Organic Chemistry tutors
- Name the epimers of d-glucose.
- Predict the products obtained when d-galactose reacts with each reagent. (b) NaOH, H2O
- Except for the Tollens test, basic aqueous conditions are generally avoided with sugars because they lead to f...
- Use Figure 23-3 <IMAGE> (the D family of aldoses) to name the following aldoses.(a) the C2 epimer of D-a...
- Which of the following molecule pairs are epimers?(a) <IMAGE>(b) <IMAGE>(c) <IMAGE>
- c. What sugar is the C-4 epimer of l-gulose?
- d. What sugar is the C-4 epimer of d-lyxose?
- What sugar is the C-3 epimer of d-fructose?
- When glucose undergoes base-catalyzed isomerization in the absence of the enzyme, mannose is oneof the product...
- (a) Draw D-allose, the C3 epimer of glucose. (b) Draw D-talose, the C2 epimer of d-galactose

